Chapter: 11th Physics : UNIT 8 : Heat and Thermodynamics
Laws of Heat Transfer
LAWS
OF HEAT TRANSFER
Prevost theory of heat exchange
Every
object emits heat radiations at all finite temperatures (except 0 K) as well as
it absorbs radiations from the surroundings. For example, if you touch someone,
they might feel your skin as either hot or cold.
A body at high temperature radiates more heat to the surroundings than it receives from it. Similarly, a body at a lower temperature receives more heat from the surroundings than it loses to it.
Prevost applied the idea of ŌĆśthermal equilibriumŌĆÖ to
radiation. He suggested that all bodies radiate energy but hot bodies radiate
more heat than the cooler bodies. At one point of time the rate of exchange of
heat from both the bodies will become the same. Now the bodies are said to be
in ŌĆśthermal equilibriumŌĆÖ.
Only
at absolute zero temperature a body will stop emitting. Therefore Prevost theory states that all bodies emit thermal radiation at all temperatures above
absolute zero irrespective of the nature of the surroundings.
Stefan Boltzmann law
Stefan
Boltzmann law states that, the total amount of heat radiated per second per
unit area of a black body is directly proportional to the fourth power of its
absolute temperature.
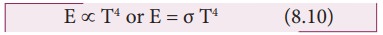
Where, Žā is known as StefanŌĆÖs constant. Its value is 5.67 ├Ś 10ŌłÆ8 W mŌłÆ2 kŌłÆ4
WienŌĆÖs displacement law
In
the universe every object emits radiation. The wavelengths of these radiations
depend on the objectŌĆÖs absolute temperature. These radiations have different
wavelengths and all the emitted wavelengths will not have equal intensity.
![]()
![]() WienŌĆÖs law states that, the
wavelength of maximum intensity of
emission of a black body radiation is inversely proportional to the absolute
temperature of the black body.
WienŌĆÖs law states that, the
wavelength of maximum intensity of
emission of a black body radiation is inversely proportional to the absolute
temperature of the black body.
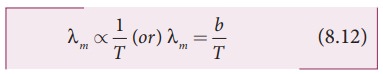
Where,
b is known as WienŌĆÖs constant. Its value is 2.898├Ś 10-3 m K
It
implies that if temperature of the body increases, maximal intensity wavelength
( ╬╗m ) shifts towards lower wavelength
(higher frequency) of electromagnetic spectrum. It is shown in Figure 8.13
Graphical representation
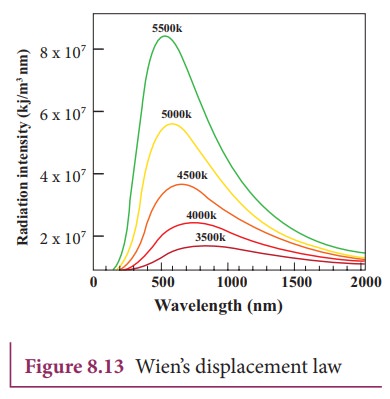
From
the graph it is clear that the peak of the wavelengths is inversely
proportional to temperature. The curve is known as ŌĆśblack body radiation
curveŌĆÖ.
WienŌĆÖs law and Vision:
Why our eye is sensitive to only visible wavelength (in the range 400 nm to 700nm)?
The
Sun is approximately taken as a black body. Since any object above 0 K will
emit radiation, Sun also emits radiation. Its surface temperature is about 5700
K. By substituting this value in the equation (8.12),
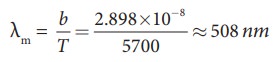
It
is the wavelength at which maximum intensity is 508nm. Since the SunŌĆÖs temperature is around 5700K, the spectrum of
radiations emitted by Sun lie between 400 nm to 700 nm which is the visible
part of the spectrum. It is shown in Figure 8.14
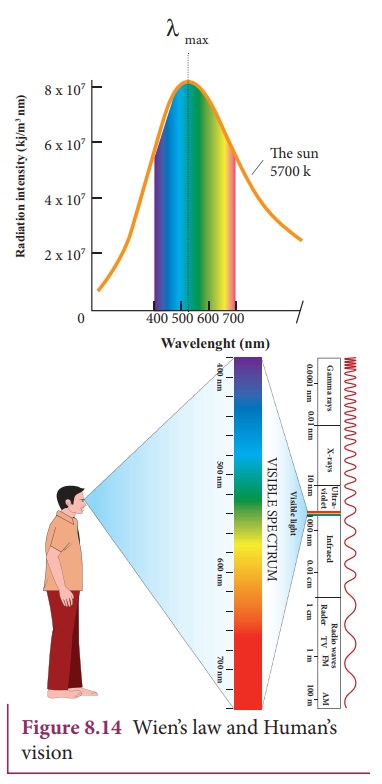
The
humans evolved under the Sun by receiving its radiations. The human eye is
sensitive only in the visible not in infrared or X-ray ranges in the spectrum.
![]()
![]() Suppose if humans had evolved in a planet near the star
Sirius (9940K), then they would have had the ability to see the Ultraviolet
rays!
Suppose if humans had evolved in a planet near the star
Sirius (9940K), then they would have had the ability to see the Ultraviolet
rays!
EXAMPLE 8.9
The
power radiated by a black body A is EA and the maximum energy
radiated was at the wavelength ╬╗A. The power radiated by another
black body B is EB = N EA and the radiated energy was at
the maximum The power radiated by a black body A is EA and the maximum energy
radiated was at the wavelength ╬╗A. The power radiated by another
black body B is EB = NEA and the radiated energy was at
the maximum wavelength, 1/2 ╬╗A. What is the value of N?
According
to2WienŌĆÖs displacement law
╬╗max
T = constant for both object A and B

Object
B has emitted at lower wavelength compared to A. So the object B would have
emitted more energetic radiation than A.
Related Topics