Physics - Change of state | 11th Physics : UNIT 8 : Heat and Thermodynamics
Chapter: 11th Physics : UNIT 8 : Heat and Thermodynamics
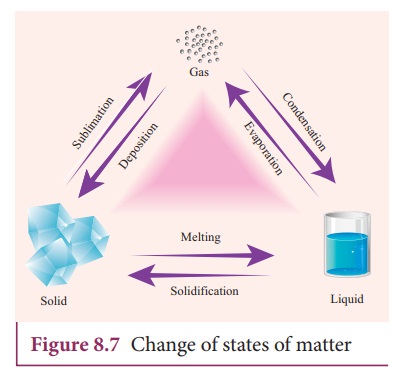
Change of state
Change
of state
All
matter exists normally in three states as solids, liquids or gases. Matter can
be changed from one state to another either by heating or cooling.
Examples:
1.
Melting (solid to liquid)
2.
Evaporation (liquid to gas)
3.
Sublimation (solid to gas)
4.
Freezing / Solidification (liquid to solid)
5.
Condensation (gas to liquid)
Latent heat capacity:
While
boiling a pot of water, the temperature of the water increases until it reaches
100 ˚C which is the boiling point of water, and then the temperature remains
constant until all the water changes from liquid to gas. During this process
heat is continuously added to the water. But the temperature of water does not
increase above its boiling point. This is the concept of latent heat capacity.
Latent heat capacity of a substance
is defined as the amount of heat energy required to change the state of a unit
mass of the material.
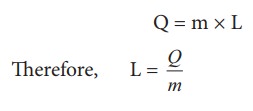
Where
L = Latent heat capacity of the substance
Q
= Amount of heat
m
= mass of the substance
The
SI unit for Latent heat capacity is J kg-1
.
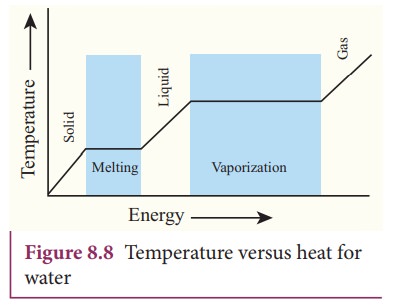
ŌĆóŌĆā The latent heat for a solid - liquid
state change is called the latent heat of fusion (Lf)
ŌĆóŌĆā The latent heat for a liquid - gas
state change is called the latent heat of vaporization (Lv)
ŌĆóŌĆā The latent heat for a solid - gas
state change is called the latent heat of
sublimation (Ls)
Triple point
the triple point of a substance is
the temperature and pressure at which the three phases (gas, liquid and solid)
of that substance coexist in thermodynamic equilibrium.
The
triple point of water is at 273.1 K and a partial vapour pressure of 611.657
Pascal.
Related Topics