Chapter: 11th Physics : UNIT 8 : Heat and Thermodynamics
Internal Energy (U)
INTERNAL
ENERGY (U)
The
internal energy of a thermodynamic system is the sum of kinetic and potential
energies of all the molecules of the system with respect to the center of mass
of the system.
The
energy due to molecular motion including translational, rotational and
vibrational motion is called internal kinetic energy (EK)
The
energy due to molecular interaction is called internal potential energy (EP).
Example: Bond energy.
U
= EK + EP
┬Ę
Since
ideal gas molecules are assumed to have no interaction with each other the
internal energy consists of only kinetic energy part (EK) which
depends on the temperature, number of particles and is independent of volume.
However this is not true for real gases like Van der Waals gases.
┬Ę
Internal
energy is a state variable. It depends only on the initial and final states of
the thermodynamic system. For example, if the temperature of water is raised
from 30┬░C to 40┬░C either by heating or by stirring, the final internal energy
depends only on the final temperature 40┬░C and not the way it is arrived at.
EXAMPLE 8.10
When
you mix a tumbler of hot water with one bucket of normal water, what will be
the direction of heat flow? Justify.
The
water in the tumbler is at a higher temperature than the bucket of normal
water. But the bucket of normal water has larger internal energy than the hot
water in the tumbler. This is because the internal energy is an extensive
variable and it depends on the size or mass of the system.
Even
though the bucket of normal water has larger internal energy than the tumbler
of hot water, heat will flow from water in the tumbler to the water in the
bucket. This is because heat flows from a body at higher temperature to the one
at lower temperature and is independent of internal energy of the system.
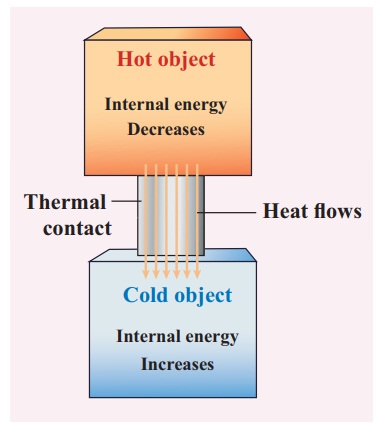
Once
the heat is transferred to an object it becomes internal energy of the object.
The right way to say is ŌĆśobject has certain amount of internal energyŌĆÖ. Heat is
one of the ways to increase the internal energy of a system as shown in the
Figure.
JouleŌĆÖs mechanical equivalent of heat
The temperature of an object can be increased by heating it or by doing some work on it. In the eighteenth century, James Prescott Joule showed that mechanical energy can be converted into internal energy and vice versa. In his experiment, two masses were attached with a rope and a paddle wheel as shown in Figure 8.19. When these masses fall through a distance h due to gravity, both the masses lose potential energy equal to 2mgh. When the masses fall, the paddle wheel turns. Due to the turning of wheel inside water, frictional force comes in between the water and the paddle wheel.
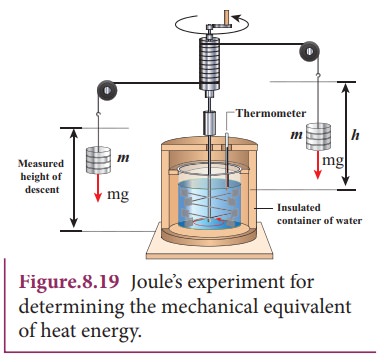
This causes a
rise in temperature of the water. This implies that gravitational potential
energy is converted to internal energy of water. The temperature of water
increases due to the work done by the masses. In fact, Joule was able to show
that the mechanical work has the same effect as giving heat. He found that to
raise 1 g of an object by 1┬░C , 4.186 J of energy is required. In earlier days
the heat was measured in calorie.
1
cal = 4.186 J
This
is called JouleŌĆÖs mechanical equivalent of heat.
EXAMPLE 8.11
A
student had a breakfast of 200 food calories. He thinks of burning this energy
by drawing water from the well and watering the trees in his school. Depth of
the well is about 25 m. The pot can hold 25L of water and each tree requires
one pot of water. How many trees can he water? (Neglect the mass of the pot and
the energy spent by walking. Take g =10 m s-2)

Solution
To
draw 25 L of water from the well, the student has to do work against gravity by
burning his energy.
Mass
of the water = 25 L = 25 kg (1L=1kg )
The
work required to draw 25 kg of water = gravitational potential energy gained by
water.
W = mgh = 25├Ś10├Ś25 = 6250 J
The total energy gained from the food = 200 food cal =200
kcal.
=
200├Ś103 ├Ś 4.186 J = 8.37
├Ś105 J
If
we assume that by using this energy the student can drawn ŌĆśnŌĆÖ pots of water
from the well, the total energy spent by him = 8.37 ├Ś 105 J = nmgh
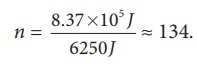
This
n is also equal to the number of trees that he can water.
Is
it possible to draw 134 pots of water from the well just by having breakfast?
No. Actually the human body does not convert entire food energy into work. It
is only approximately 20% efficient. It implies that only 20% of 200 food
calories is used to draw water from the well. So 20 % of the 134 is only 26
pots of water. It is quite meaningful. So he can water only 26 trees.
The
remaining energy is used for blood circulation and other functions of the body.
It is to be noted that some energy is always ŌĆśwastedŌĆÖ. Why is it that the body
cannot have 100% efficiency? You will find the answer in section 8.9
First law of thermodynamics
The
first law of thermodynamics is a statement of the law of conservation of
energy. In Newtonian mechanics conservation of energy involves kinetic and
potential energies of bulk objects. But the first law of thermodynamics
includes heat also. This law states that ŌĆśChange
in internal energy (ΔU) of the system
is equal to heat supplied to the system (Q) minus the work done by the system (W)
on the surroundingsŌĆÖ. Mathematically it on
the surroundingsŌĆÖ. Mathematically it is
written as

The
internal energy of a thermodynamic system can be changed either by heating or
by work as shown below.
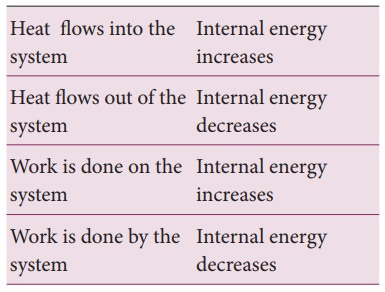
Based
on the above table the sign convention is introduced to use first law of
thermodynamics appropriately. It is shown in the following table and the Figure
8.20.
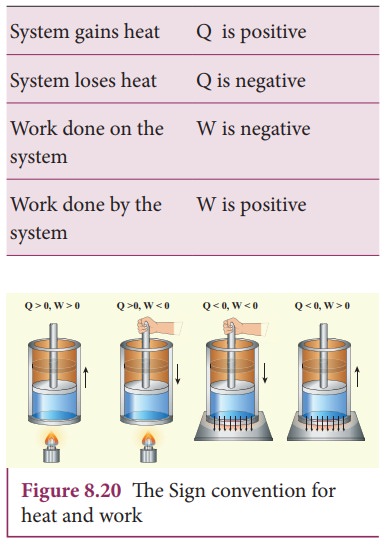
Even
though we often explain first law of thermodynamics using gases, this law is
universal and applies to liquids and solids also.
EXAMPLE 8.12
A
person does 30 kJ work on 2 kg of water by stirring using a paddle wheel. While
stirring, around 5 kcal of heat is released from water through its container to
the surface and surroundings by thermal conduction and radiation. What is the
change in internal energy of the system?
Solution
Work
done on the system (by the person while stirring), W = -30 kJ = -30,000J
Heat
flowing out of the system,
Q
= -5 kcal = 5 ├Ś 4184 J =-20920 J
Using
First law of thermodynamics
ŌłåU
= Q-W
ŌłåU
= -20,920 J-(-30,000) J
ŌłåU
= -20,920 J+30,000 J = 9080 J
Here,
the heat lost is less than the work done on the system, so the change in
internal energy is positive.
EXAMPLE 8.13
Jogging
every day is good for health. Assume that when you jog a work of 500 kJ is done
and 230 kJ of heat is given off. What is the change in internal energy of your
body?
Solution

Work
done by the system (body),
W
= +500 kJ
Heat
released from the system (body),
Q
= ŌĆō230 kJ
The
change in internal energy of a body
=
╬öU= ŌĆō 230 kJ ŌĆō 500 kJ = ŌĆō 730 kJ
Quasi-static process
Consider
a system of an ideal gas kept in a cylinder of volume V at pressure P and
temperature T. When the piston attached to the cylinder moves outward the
volume of the gas will change. As a result the temperature and pressure will
also change because all three variables P,T and V are related by the equation
of state PV = NkT. If a block of some mass is kept on the piston, it will
suddenly push the piston downward. The pressure near the piston will be larger
than other parts of the system. It implies that the gas is in non-equilibrium
state. We cannot determine pressure, temperature or internal energy of the
system until it reaches another equilibrium state. But if the piston is pushed
very slowly such that at every stage it is still in equilibrium with
surroundings, we can use the equation of state to calculate the internal
energy, pressure or temperature. This kind of process is called quasi-static
process.
A
quasi-static process is an infinitely slow process in which the system changes
its variables (P,V,T) so slowly such that it remains in thermal, mechanical and
chemical equilibrium with its surroundings throughout. By this infinite slow
variation, the system is always almost close to equilibrium state.
EXAMPLE 8.14
Give
an example of a quasi-static process. Consider a container of gas with volume
V, pressure P and temperature T. If we add sand particles one by one slowly on
the top of the piston, the piston will move inward very slowly. This can be
taken as almost a quasi-static process. It is shown in the figure
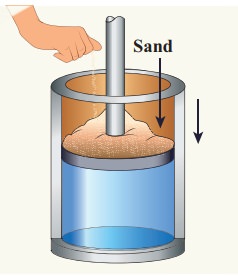
Sand
particles added slowly- quasi-static process
Work done in volume changes
Consider
a gas contained in the cylinder fitted with a movable piston. Suppose the gas
is expanded quasi-statically by pushing the piston by a small distance dx as
shown in Figure 8.21. Since the expansion occurs quasi-statically the pressure,
temperature and internal energy will have unique values at every instant.
The
small work done by the gas on the piston

The
force exerted by the gas on the piston F = PA. Here A is area of the piston and
P is pressure exerted by the gas on the piston.
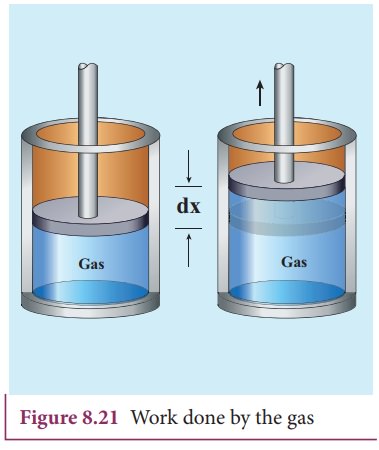
Equation
(8.14) can be rewritten as

But
Adx = dV= change in volume during this expansion process.
So
the small work done by the gas during the expansion is given by

Note
here that is positive since the volume is increased. Here, is positive.
In
general the work done by the gas by increasing the volume from Vi to Vf is given by

Suppose if the work is done on the system, then Vi > Vf . Then, W is negative.
Note
here the pressure P is inside the integral in equation (8.17). It implies that
while the system is doing work, the pressure need not be constant. To evaluate
the integration we need to first express the pressure as a function of volume
and temperature using the equation of state.
PV diagram
PV
diagram is a graph between pressure P and volume V of the system. The P-V
diagram is used to calculate the amount of work done by the gas during
expansion or on the gas during compression. In Unit 2, we have seen that the
area under the curve will give integration of the function from lower limit to
upper limit. The area under the PV diagram will give the work done during
expansion or compression which is shown in Figure 8.22
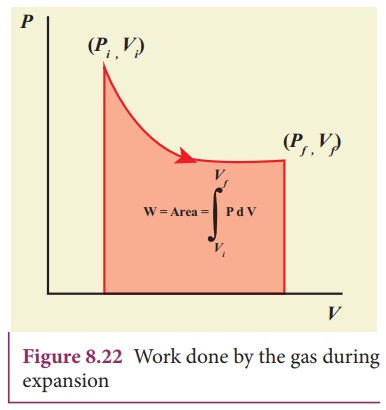
The
shape of PV diagram depends on the nature of the thermodynamic process.
EXAMPLE 8.15
A
gas expands from volume 1m3 to 2m3 at constant atmospheric pressure.
(a) Calculate the work done
by the gas.
(b) Represent the work done in PV diagram.
Solution
(a) The pressure P = 1 atm =
101 kPa, Vf =2 m3 and V1 = 1m3
From equation (8.17) W =
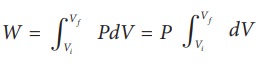
Since
P is constant. It is taken out of the integral.
W = P (Vf ŌĆō Vi) =
101├Ś103 ├Ś (2 ŌĆō 1) = 101 kJ
(b) Since the pressure is
kept constant, PV diagram is straight line as shown in the figure. The area is equal
to work done by the gas.
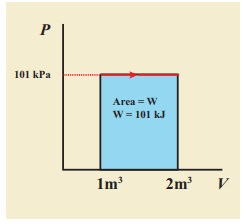
Note
the arrow mark in the curve. Suppose the work is done on the system, then
volume will decreases and the arrow will point in the opposite direction.
Related Topics