Electrostatics - Coulomb’s Law | 12th Physics : Electrostatics
Chapter: 12th Physics : Electrostatics
Coulomb’s Law
COULOMB’S LAW
In the year 1786,
Coulomb deduced the expression for the force between two stationary point
charges in vacuum or free space. Consider two point charges q1 and q2
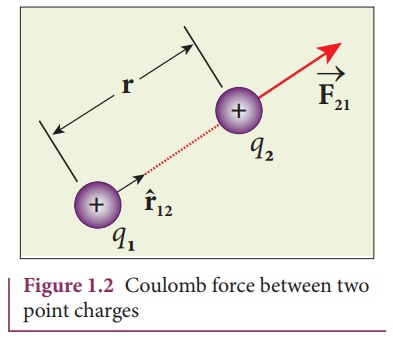
at rest in vacuum, and separated by a distance of r, as shown in Figure 1.2.
According to Coulomb, the force on the point charge q2 exerted by
another point charge q1 is
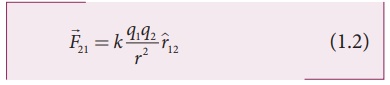
where ^r12
is the unit vector directed from charge q1 to charge q2
and k is the proportionality constant.
Important aspects of Coulomb’s law
(i) Coulomb's law states
that the electrostatic force is directly proportional to the product of the
magnitude of the two point charges and is inversely proportional to the square
of the distance between the two point charges.
(ii) The force on the
charge q2 exerted by the charge q1 always lies along the
line joining the two charges. ^r12 is the unit vector
pointing from charge q1 to q2. It is shown in the Figure
1.2. Likewise, the force on the charge q1 exerted by q2 is along -r^12 (i.e., in the
direction opposite to r^12 ).
(iii) In SI units, 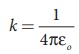 and its value is 9 × 109
N m2 C-2. Here ε is the permittivity of free space or
vacuum and the value of
and its value is 9 × 109
N m2 C-2. Here ε is the permittivity of free space or
vacuum and the value of
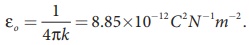
(iv) The magnitude of
the electrostatic force between two charges each of one coulomb and separated
by a distance of 1 m is calculated as follows:
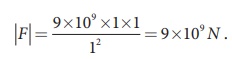
This is a huge quantity,
almost equivalent to the weight of one million ton. We never come across 1
coulomb of charge in practice. Most of the electrical phenomena in day-to-day
life involve electrical charges of the order of µC (micro coulomb) or nC (nano
coulomb).
(v) In SI units,
Coulomb’s law in vacuum takes the form 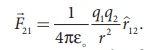 In a medium of
permittivity ε, the force between two point charges is given by
In a medium of
permittivity ε, the force between two point charges is given by 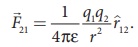
Since ε>εo
, the force between two point charges in
a medium other than vacuum is always less than that in vacuum. We define the
relative permittivity for a given medium as εr>ε/εo . For
vacuum or air, εr =
1 and ε for all other media εr>
1.![]()
(vi) Coulomb’s law has same structure as Newton’s law of gravitation. Both
are inversely proportional to the square of the distance between the particles.
The electrostatic force is directly proportional to the product of the
magnitude of two point charges and gravitational force is directly proportional
to the product of two masses. But there are some important differences between
these two laws.
·
The gravitational force between two masses is always attractive
but Coulomb force between two charges can be attractive or repulsive, depending
on the nature of charges.
·
The value of the gravitational constant G = 6.626 × 10-11
N m2 kg-2. The value of the constant k in Coulomb law is
k = 9 × 109 N m2 C-2. Since k is much more
greater than G, the electrostatic force is always greater in magnitude than
gravitational force for smaller size objects.
·
The gravitational force between two masses is independent of the
medium. For example, if 1 kg of two masses are kept in air or inside water, the
gravitational force between two masses remains the same. But the electrostatic
force between the two charges depends on nature of the medium in which the two
charges are kept at rest.
·
The gravitational force between two point masses is the same
whether two masses are at rest or in motion. If the charges are in motion, yet
another force (Lorentz force) comes into play in addition to coulomb force.
(vii) The force on a
charge q1 exerted by a point charge q2 is given by
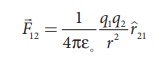
Here ^r21
is the unit vector from charge q2 to q1.
But ^r21
= − ^r12 ,
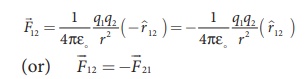
Therefore, the
electrostatic force obeys Newton’s third law.
(viii) The expression
for Coulomb force is true only for point charges. But the point charge is an
ideal concept. However we can apply Coulomb’s law for two charged objects whose
sizes are very much smaller than the distance between them. In fact, Coulomb
discovered his law by considering the charged spheres in the torsion balance as
point charges. The distance between the two charged spheres is much greater
than the radii of the spheres.
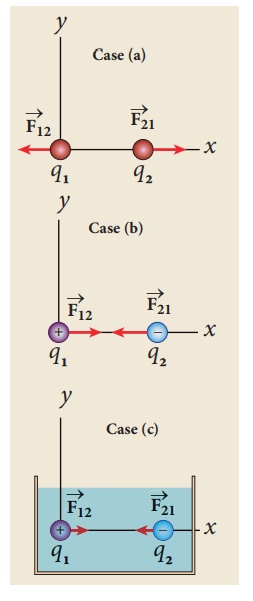
EXAMPLE 1.2
Consider two point
charges q1 and q2 at rest as shown in the figure.
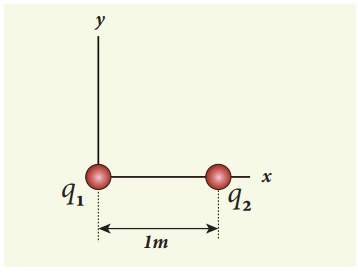
They are separated by a
distance of 1m. Calculate the force experienced by the two charges for the
following cases:
(a) q1 = +2μC
and q2 = +3μC
(b) q1 = +2μC
and q2 = -3μC
(c) q1= +2μC
and q2 = -3μC kept in water (εr = 80)
Solution



EXAMPLE 1.3
Two small-sized
identical equally charged spheres, each having mass 1 mg are hanging in
equilibrium as shown in the figure. The length of each string is 10 cm and the
angle θ is 7° with the vertical. Calculate the magnitude of the charge in each
sphere.
(Take g = 10 ms−2)
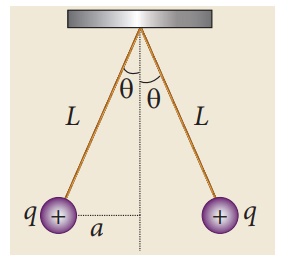
Solution
If the two spheres are
neutral, the angle between them will be 0o when hanged vertically. Since they
are positively charged spheres, there will be a repulsive force between them
and they will be at equilibrium with each other at an angle of 7° with the
vertical. At equilibrium, each charge experiences zero net force in each
direction. We can draw a free body diagram for one of the charged spheres and
apply Newton’s second law for both vertical and horizontal directions.
The free body diagram is
shown below.
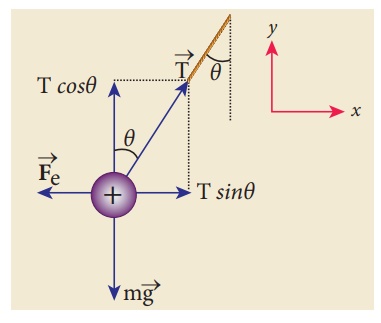
In the x-direction, the acceleration of the charged sphere is zero.
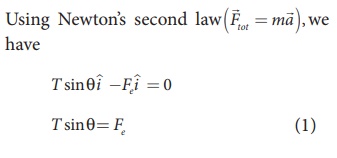
Here T is the tension acting on the
charge due to the string and Fe is the electrostatic force between
the two charges.
In the y-direction also, the net
acceleration experienced by the charge is zero.

EXAMPLE 1.4
Calculate the
electrostatic force and gravitational force between the proton and the electron
in a hydrogen atom. They are separated by a distance of 5.3 × 10-11
m. The magnitude of charges on the electron and proton are 1.6 × 10-19
C. Mass of the electron is me = 9.1 × 10-31 kg and mass
of proton is mp = 1.6 × 10-27 kg.
Solution
The proton and the
electron attract each other. The magnitude of the electrostatic force between
these two particles is given by
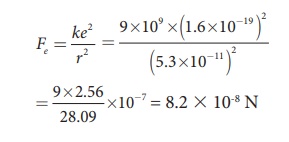
The gravitational force between the
proton and the electron is attractive. The magnitude of the gravitational force
between these particles is

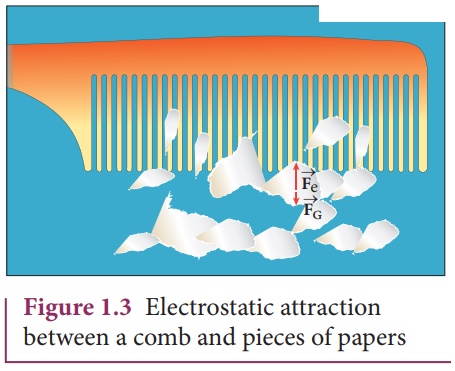
The electrostatic force between a
proton and an electron is enormously greater than the gravitational force
between them. Thus the gravitational force is negligible when compared with the
electrostatic force in many situations such as for small size objects and in
the atomic domain. This is the reason why a charged comb attracts an uncharged
piece of paper with greater force even though the piece of paper is attracted
downward by the Earth. This is shown in Figure 1.3
1. Superposition principle
Coulomb’s law explains
the interaction between two point charges. If there are more than two charges,
the force on one charge due to all the other charges needs to be calculated.
Coulomb’s law alone does not give the answer. The superposition principle
explains the interaction between multiple charges.
According to this
superposition principle, the total force
acting on a given charge is equal to the vector sum of forces exerted on it by
all the other charges.
Consider a system of n
charges, namely q1, q2, q3 ….qn.
The force on q1 exerted by the charge q2
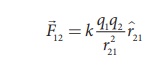
Here ^r21
is the unit vector from q2 to q1 along the line joining
the two charges and r21 is the distance between the charges q1
and q2. The electrostatic force between two charges is not affected
by the presence of other charges in the neighbourhood.
The force on q1
exerted by the charge q3 is
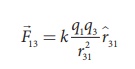
By continuing this, the
total force acting on the charge q1 due to all other charges is
given by
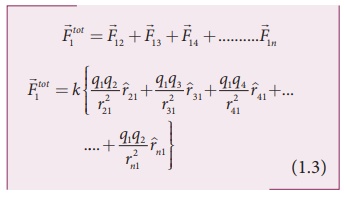
EXAMPLE 1.5
Consider four equal
charges q1,q2, q3 and q4 = q = +1μC
located at four different points on a circle of radius 1m, as shown in the
figure. Calculate the total force acting on the charge q1 due to all
the other charges.
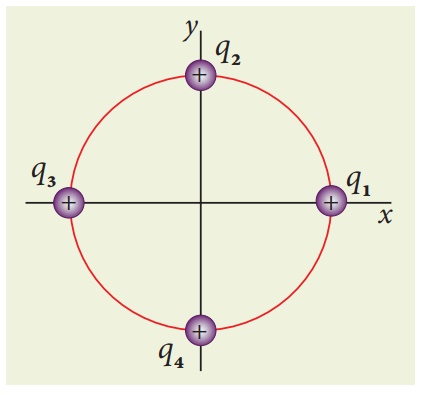
Solution
According to the
superposition principle, the total electrostatic force on charge q1 is the
vector sum of the forces due to the other charges,
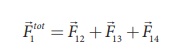
The following diagram
shows the direction of each force on the charge q1.
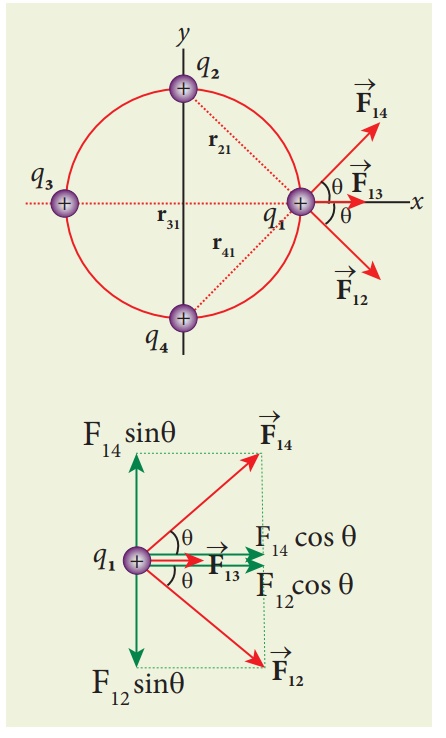
The charges q2
and q4 are equi-distant from q1. As a result the
strengths (magnitude) of the forces ![]() and
and ![]() are
the same even though their directions are different. Therefore the vectors
representing these two forces are drawn with equal lengths. But the charge q3
is located farther compared to q2 and q4. Since the
strength of the electrostatic force decreases as distance increases, the
strength of the force
are
the same even though their directions are different. Therefore the vectors
representing these two forces are drawn with equal lengths. But the charge q3
is located farther compared to q2 and q4. Since the
strength of the electrostatic force decreases as distance increases, the
strength of the force ![]() is lesser than that of forces
is lesser than that of forces ![]() and
and ![]() . Hence the vector representing the force
. Hence the vector representing the force ![]() is drawn with smaller length compared to that for forces
is drawn with smaller length compared to that for forces ![]() and
and ![]() .
.
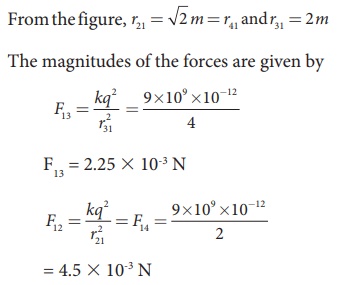
From the figure, the
angle θ = 45º. In terms of the components, we have
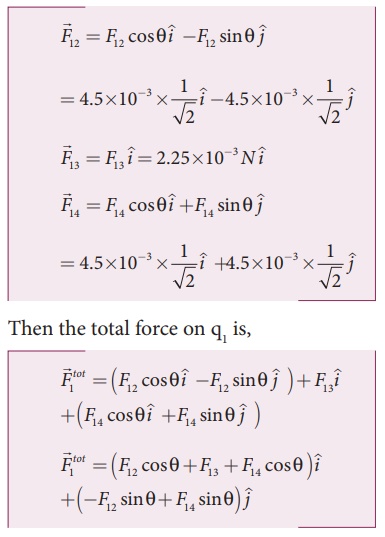
Since F12 = F14,
the jth component is zero.
Hence we have
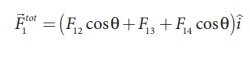
substituting the values
in the above equation,
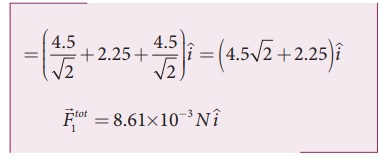
The resultant force is
along the positive x axis.
Related Topics