Biot - Savart Law | Physics - Magnetic dipole moment of revolving electron | 12th Physics : Magnetism and Magnetic Effects of Electric Current
Chapter: 12th Physics : Magnetism and Magnetic Effects of Electric Current
Magnetic dipole moment of revolving electron
Magnetic dipole moment of revolving electron
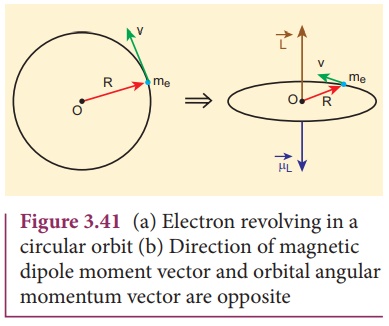
Suppose an electron undergoes circular motion around the nucleus as shown in Figure 3.41. The circulating electron in a loop is like current in a circular loop (since flow of charge is current). The magnetic dipole moment due to current carrying circular loop is

In magnitude,
µL = I A
If T is the time period of an electron, the current due to circular motion of the electron is

where −e is the charge of an electron. If R is the radius of the circular orbit and v is the velocity of the the velocity of the electron in the circular orbit, then

Using equation (3.45) and equation (3.46) in equation (3.44), we get
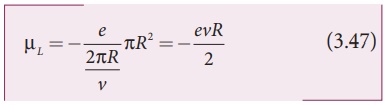
where A = πR2 is the area of the circular loop. By definition, angular momentum of the electron about O is
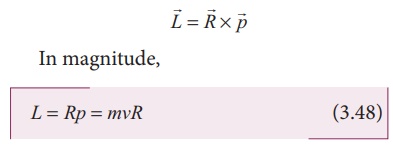
Using equation (3.47) and equation (3.48), we get
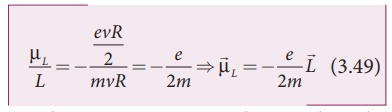
The negative sign indicates that the magnetic moment and angular momentum are in opposite direction.
In magnitude,
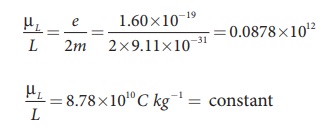
The ratio µL/L is a constant and also known as gyro-magnetic ratio (e/2m). It must be noted that the gyro-magnetic ratio is a constant of proportionality which connects angular momentum of the electron and the magnetic moment of the electron.
According to Neil’s Bohr quantization rule, the angular momentum of an electron moving in a stationary orbit is quantized, which means,
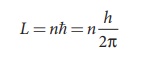
where, h is the Planck’s constant (h = 6.63 x 10-34 J s ) and number n takes natural numbers
(i.e., n = 1,2,3,....). Hence,

The minimum magnetic moment can be obtained by substituting n = 1,
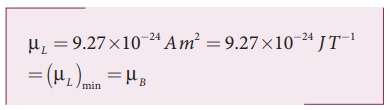
where, µB =eh/4πm= 9.27 ×10−24 Am2 is called Bohr magneton. This is a convenient unit with which one can measure atomic magnetic moments.
Note: Bohr quantization rule will be discussed in unit 8 of second volume
Related Topics