Chapter: 11th Physics : UNIT 7 : Properties of Matter
Surface tension: Angle of contact
Angle
of contact
When the free surface of a liquid comes in contact with a solid, then the surface of the liquid becomes curved at the point of contact. Whenever the liquid surface becomes a curve, then the angle between the two medium (solid-liquid interface) comes in the picture. For an example, when a glass plate is dipped in water with sides vertical as shown in figure, we can observe that the water is drawn up to the plate. In the same manner, instead of water the glass plate is dipped in mercury, the surface is curved but now the curve is depressed as shown in Figure 7.29.
The angle between the tangent to the liquid surface at the
point of contact and the solid surface inside the liquid is known as the angle of contact between the solid and the
liquid. It is denoted by ╬Ė (Read it
as ŌĆ£thetaŌĆØ which is Greek alphabet
small letter).
Its value is different at interfaces of different pairs of solids and liquids. In fact, it is the factor which decides whether a liquid will spread on the surface of a chosen solid or it will form droplets on it.
Let
us consider three interfaces such as liquid-air, solid-air and solid-liquid
with reference to the point of contact ŌĆśOŌĆÖ and the interfacial surface tension
forces Tsa, Tsl and Tla on the respective
interfaces as shown in Figure 7.26.

Since
the liquid is stable under equilibrium, the surface tension forces between the
three interfaces must also be in equilibrium. Therefore,
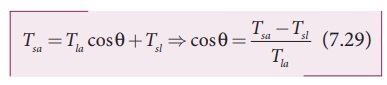
From
the above equation, there are three different possibilities which can be
discussed as follows.
(i)
If Tsa > Tsl and Tsa ŌłÆ Tsl >
0 (water-plastic interface) then the angle of contact ╬Ė is acute angle (╬Ė less
than 90┬░) as cos╬Ė is positive.
(ii)
If Tsa < Tsl and Tsa ŌłÆ Tsl <
0 (water-leaf interface) then the angle of contact is obtuse angle (╬Ė less than
180┬░) and as cos╬Ė is negative.
(iii)
If Tsa > Tla + Tsl then there will be no
equilibrium and liquid will spread over the solid.
Therefore,
the concept of angle of contact between the solid-liquid interface leads to
some practical applications in real life. For example, soaps and detergents are
wetting agents. When they are added to an aqueous solution, they will try to
minimize the angle of contact and in turn penetrate well in the cloths and
remove the dirt. On the other hand, water proofing paints are coated on the
outer side of the building so that it will enhance the angle of contact between
the water and the painted surface during the rainfall.
Related Topics