Properties of Matter | Physics - Short Questions and Answer | 11th Physics : UNIT 7 : Properties of Matter
Chapter: 11th Physics : UNIT 7 : Properties of Matter
Short Questions and Answer
Properties of Matter (Physics)
Short Answer Questions
1. Define stress and strain.
ŌŚÅ The force per unit area is called as stress, Stress Žā = Force / Area = F / A .
ŌŚÅ The SI unit of stress is N mŌłÆ2 or Pascal (Pa) and its dimension is [MLŌłÆ2TŌłÆ2].
ŌŚÅ The fractional chance in the size of the object or strain measures the degree of deformation.
ŌŚÅ Stain, ╬Ą = Change in size / Original size = ŌłåØæÖ / ØæÖ
2. State HookeŌĆÖs law of elasticity.
For a small deformation, within the elastic limit, the stress and strain are proportional to each other.
3. Define PoissonŌĆÖs ratio.
ŌŚÅ The ratio of relative contraction (lateral strain) to relative expansion (longitudinal strain).
ŌŚÅ It is denoted by the symbol ╬╝ .
ŌŚÅ Poisson's ratio, ╬╝ = lateral strain / longitudinal strain
4. Explain elasticity using intermolecular forces.
ŌŚÅ In a solid, Inter-atomic forces bind two or more atoms together and the atoms occupy the positions of stable equilibrium.
ŌŚÅ When a deforming force is applied on a body. Its atoms are pulled apart or pushed closer.
ŌŚÅ When the deforming force is removed, inter-atomic forces of attraction or repulsion restore the atoms to their equilibrium positions.
ŌŚÅ If a body regains it original shape and size after the removal of deforming force, it is said to be elastic and the property is called elasticity.
5. Which one of these is more elastic, steel or rubber? Why?
ŌŚÅ Steel is more elastic than rubber because the steel has higher young's modulus than rubber.
ŌŚÅ If an equal stress is applied to both steel and rubber, the steel produces less strain.
6. A spring balance shows wrong readings after using for a long time. Why?
When the spring balance have been used for a long time they develop elastic fatigue in them and therefore the reading shown by such balances will be wrong.
7. What is the effect of temperature on elasticity?
If the temperature of the substance increases, its elasticity decreases.
8. Write down the expression for the elastic potential energy of a stretched wire.
ŌŚÅ Consider a wire whose un-stretch length is L and area of cross section is A.
ŌŚÅ Let a force produce an extension l and the elastic limit of the wire has not been exceeded and there is no loss in energy.
ŌŚÅ Then, the work done by the force F is equal to the energy gained by the wire.
ŌŚÅ The work done in stretching the wire by dl, dW = F dl
ŌŚÅ The total work done in stretching the wire from 0 to l is W =╩ā0l Fdl ------ (1)
ŌŚÅ From young's modulus of elasticity, Y = [F/A] ├Ś [L/l] ŌćÆ F = YAl / L ------ (2)
ŌŚÅ Substituting equation (2) in equation (1), we get W= ╩ā0l [YAl / L] dl
ŌŚÅ l is the dummy variable, so l to lŌĆÖ
ŌŚÅ 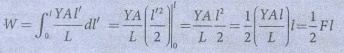
W = ┬Į Fl = Elastic potential energy.
9. State PascalŌĆÖs law in fluids.
If the pressure in a liquid is changed at a particular point, the change is transmitted to the entire liquid without being diminished in magnitude.
10. State Archimedes principle.
It states that when a body is partially or wholly immersed in a fluid, it experiences an upward thrust equal to weight of the fluid displaced by it and its up-thrust acts through the centre of gravity of the liquid displaced.
11. What do you mean by upthrust or buoyancy?
The upward force exerted by a fluid that opposes the weight of an immersed object in a fluid is called up-thrust or buoyant force and the phenomenon is called buoyancy.
12. State the law of floatation.
The law of floatation states that a body will float in a liquid if the weight of the liquid displaced by the immersed part of the body equal to the weight of the body.
13. Define coefficient of viscosity of a liquid.
The coefficient of viscosity is defined as the force of viscosity acting between two layers per unit area and unit velocity gradient of the liquid. Its unit Nsm-2 and dimension is [ML-1T-1].
14. Distinguish between streamlined flow and turbulent flow.
ŌŚÅ Streamlined flow: When a liquid flows such that each particle of the liquid passing through a point moves along the same path with the same velocity as its predecessor then the flow of liquid is said to be a streamlined flow.
ŌŚÅ The velocity of the particle at any point is constant. It is also referred to as steady or laminar flow.
ŌŚÅ The actual path taken by the particle of the moving fluid is called a streamline, which is the curve, the tangent to which at any point gives the direction of the flow of the fluid at that point.
ŌŚÅ Turbulent flow: When the speed of the moving fluid exceeds the critical speed vc , the motion becomes turbulent.
ŌŚÅ The velocity changes both in magnitude and direction from particle to particle.
ŌŚÅ The path taken by the particles in turbulent flow becomes erratic and whirlpool-like circles called eddy current or eddies.
15. What is ReynoldŌĆÖs number? Give its significance.
ŌŚÅ Reynolds number Rc is a dimensionless number, which is used to find out the nature of flow of the liquid Rc = ŽüvD /╬Ę
Where Žü -density of the liquid. v-The velocity of flow of liquid. D - Diameter of the pipe, ╬Ę -The coefficient of viscosity of the fluid.
16. Define terminal velocity.
The maximum constant velocity acquired by a body while falling freely through a viscous medium is called the terminal velocity.
17. Write down the expression for the StokeŌĆÖs force and explain the symbols involved in it.
Viscous force F acting on a spherical body of radius r depends directly on
i) radius (r) of the sphere
ii) velocity (v) of the sphere and
iii) coefficient of viscosity ╬Ę of the liquid F = 6ŽĆ╬Ęrv
18. State BernoulliŌĆÖs theorem.
According to Bernoulli's theorem, the sum of pressure energy, kinetic energy, and potential energy per unit mass of an incompressible, non-viscous fluid in a streamlined flow remains a constant.
19. What are the energies possessed by a liquid? Write down their equations.
A liquid in a steady flow can possess three kinds of energy.
They are (1) Kinetic energy. (2) Potential energy, and (3) Pressure energy, respectively.ŌĆā
KE = ┬Į mv2 ŌĆ”ŌĆ”ŌĆ” (1)
PE = mgh ŌĆ”ŌĆ”ŌĆ”ŌĆ” (2)
F├Śd = W = PV = pressure energy ŌĆ”ŌĆ”ŌĆ” (3)
20. Two streamlines cannot cross each other. Why?
ŌŚÅ If two streamlines can not cross each other.
ŌŚÅ If they do so, the particles of the liquid at the point of intersection will have two different directions for their flow, which will destroy the steady nature of the liquid flow.
21. Define surface tension of a liquid. Mention its S.I unit and dimension.
The surface tension of a liquid is defined as the energy per unit area of the surface of a liquid. Its unit is N m-1 and dimension is [MT-2].
22. How is surface tension related to surface energy?
ŌŚÅ Consider a rectangular frame of wire ABCD in a soap solution. Let AB be the movable wire.
ŌŚÅ Suppose the frame is dipped in soap solution, soap film is formed which pulls the wire AD inwards due to surface tension.
ŌŚÅ Let F be the force due to surface tension.
ŌŚÅ Then F=(2T) ØæÖ Here, 2 is introduced because it has two free surfaces.
ŌŚÅ Suppose AB is moved by a small distance to a new position A'B'. Since the area increases, some work has to be done against the inward force due to surface tension.
ŌŚÅ Work done = Force ├Ś distance = (2T) ØæÖ (Ōłåx)
ŌŚÅ Increases in area of the film : ŌłåA = (2ØæÖ) (Ōłåx) = 2ØæÖ Ōłåx
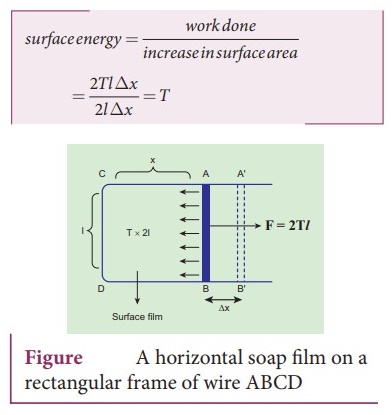
ŌŚÅ Surface energy = work done / increase in surface area = 2TØæÖŌłåx / 2ØæÖŌłåx = T
ŌŚÅ The surface energy per unit area of a surface is numerically equal to the surface tension.
23. Define angle of contact for a given pair of solid and liquid.
The angle between the tangent to the liquid surface at the point of contact and the solid surface is known as the angle of contact.
24. Distinguish between cohesive and adhesive forces.
ŌŚÅ The force between the like molecules which holds the liquid together is called 'cohesive force'.
ŌŚÅ When the liquid is in contact with a solid, the molecules of these solid and liquid will experience an attractive force which called 'adhesive force'.ŌĆā
25. What are the factors affecting the surface tension of a liquid?
1) The presence of any contamination or impurities considerably affects the force of surface tension depending upon the degree of contamination.
2) The presence of dissolved substances can also affect the value of surface tension. For example, a highly soluble substances like sodium chloride (NaCl) when dissolved in water (H2O) increases the surface tension of water. But the sparingly soluble substances like phenol or soap solution when mixed in water decreases the surface tension of water.
3) Electrification affects the surface tension. When a liquid is electrified, surface tension decreases. Since external force acts on the liquid surface due to electrification, area of the liquid surface increases which acts against the contraction phenomenon of the surface tension. Hence, it decreases.
4) Temperature plays a very crucial role in altering the surface tension of a liquid. Obviously the surface tension decreases linearly with the rise of temperature.
26. What happens to the pressure inside a soap bubble when air is blown into it?
When air is blown into a soap bubble its radius will increase. Due to this increase in radius, the excess pressure inside a soap bubble will decrease because excess pressure is inversely proportional to the radius of the bubble.
27. What do you mean by capillarity or capillary action?
The rise or fall of a liquid in a narrow tube is called capillarity or capillary action.
28. A drop of oil placed on the surface of water spreads out. But a drop of water place on oil contracts to a spherical shape. Why?
ŌŚÅ A drop of oil placed on the surface of water spreads because the force of adhesive between water and oil molecules dominates the cohesive force of oil molecules.
ŌŚÅ Cohesive force of water molecules dominates the adhesive force between water and oil molecules. So drop of water on oil contracts to spherical shape.
29. State the principle and usage of Venturimeter.
ŌŚÅ Bernoulli's theorem is the principle of Venturimeter.
ŌŚÅ Venturimeter is used to measure the rate of flow or flow speed of the incompressible fluid flowing through a pipe.
Related Topics