Chapter: Modern Analytical Chemistry: Electrochemical Methods of Analysis
Voltammetric Methods of Analysis
Voltammetric Methods of Analysis
In voltammetry a time-dependent potential is applied to an electrochemical cell, and the current
flowing through the cell is measured as a function
of that poten- tial. A plot of current as a function of applied potential is called a voltammogram and is the electrochemical equivalent of a spectrum
in spectroscopy, provid-
ing quantitative and qualitative information about the species
involved in the oxidation or reduction reaction.15 The earliest voltammetric technique to be introduced was
polarography, which was developed
by Jaroslav Heyrovsky (1890–1967) in the early 1920s, for which he was awarded the Nobel Prize in chemistry
in 1959. Since then, many different
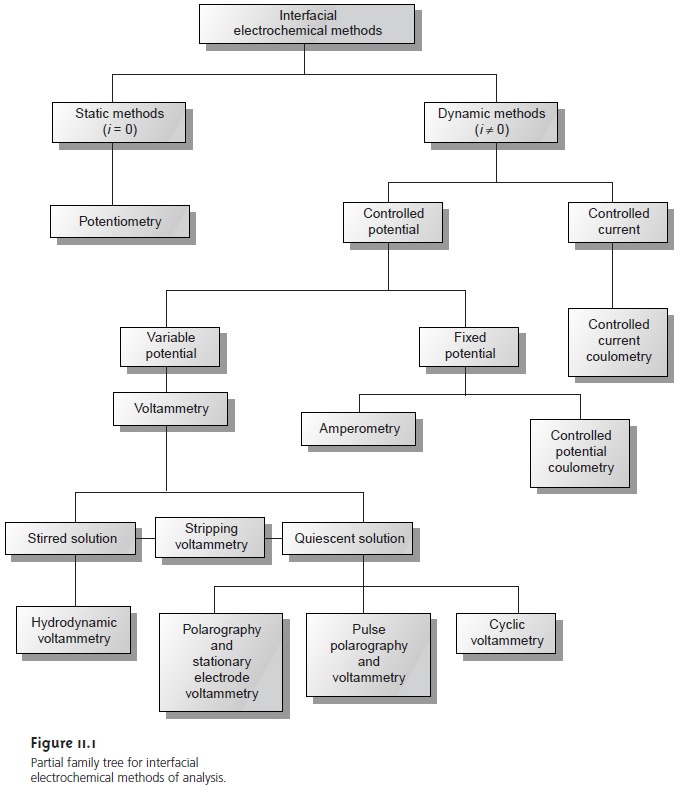
forms of voltammetry have been developed, a few of which are noted
in Figure 11.1.
Before examining these
techniques and their applications in more detail,
however, we must first consider
the basic experimental design for making voltammetric
measure- ments and the factors
influencing the shape of the resulting
voltammogram.
Related Topics