Chapter: Medical Surgical Nursing: Assessment and Management of Patients With Hematologic Disorders
Therapies for Blood Disorders
Therapies for Blood Disorders
SPLENECTOMY
The
surgical removal of the spleen (splenectomy) is sometimes necessary after
trauma to the abdomen. Because the spleen is very vascular, severe hemorrhage
can result if the spleen is ruptured. Under such circumstances, splenectomy
becomes an emergency procedure.
Splenectomy
is also a possible treatment for other hemato-logic disorders. For example, an
enlarged spleen may be the site of excessive destruction of blood cells. If the
destruction is life-threatening, surgery may be lifesaving. This is the case in
autoimmune hemolytic anemia or ITP when these disorders do not respond to more
conservative measures, such as cortico-steroid therapy. Some patients with
severe anemia due to in-herited RBC defects (eg, thalassemia) may also benefit
from splenectomy.
In
general, the mortality rate after splenectomy is low. Lap-aroscopic splenectomy
can be used in selected patients, with a resultant decrease in the
postoperative morbidity rate. Compli-cations that may result from surgery are
atelectasis, pneumonia, abdominal distention, and abscess formation. Although
young children are at the highest risk after splenectomy, all age groups are
vulnerable to overwhelming lethal infections and should re-ceive pneumovax
before undergoing this surgical procedure if possible.
Patients
are instructed to seek prompt medical attention if even relatively minor
symptoms of infection occur. Often, patients with high platelet counts have
even higher counts after splenectomy— more than 1 million/mm3—which can predispose
them to serious thrombotic or hemorrhagic problems. This increase is, however,
transient.
THERAPEUTIC APHERESIS
Apheresis
is a Greek word meaning separation. In therapeutic apheresis (or pheresis),
blood is taken from the patient and passed through a centrifuge, where a
specific component is separated from the blood and removed (Table 33-9). The
remaining blood is then returned to the patient. The entire system is closed,
so the risk of bacterial contamination is extremely low. When platelets or WBCs
are removed, the decrease in these cells within the circulation is temporary.
However, the temporary decrease provides a window of time until suppressive
medica-tions (eg, chemotherapy) can have therapeutic effects. Some-times plasma
is removed rather than blood cells—typically so that specific, abnormal
proteins within the plasma will be tran-siently lowered until a long-term
therapy can be initiated.

Apheresis
is also used to obtain larger amounts of platelets from a donor than can be
provided from a single unit of whole blood. A unit of platelets obtained in
this way is equivalent to six to eight units of platelets obtained from six to
eight separate donors via standard blood donation methods. Platelet donors can
have their platelets apheresed as often as every 14 days. WBCs can be obtained
similarly, typically after the donor has received growth factors (G-CSF,
GM-CSF) to stimulate the formation of additional WBCs and thereby increase the
WBC count. The use of these growth factors also stimulates the re-lease of stem
cells within the circulation. Apheresis is used to harvest these stem cells
(typically over a period of several days) for use in PBSCT.
THERAPEUTIC PHLEBOTOMY
Therapeutic
phlebotomy is the removal of a certain amount of blood under controlled
conditions. Patients with elevated hematocrits (eg, those with polycythemia
vera) or excessive iron absorption (eg, hemochromatosis) can usually be managed
by pe-riodically removing 1 unit (about 500 mL) of whole blood. Even-tually
this process can produce iron deficiency, leaving the patientunable to produce
as many RBCs. The actual procedure for ther-apeutic phlebotomy is similar to
that for blood donation (see later discussion).
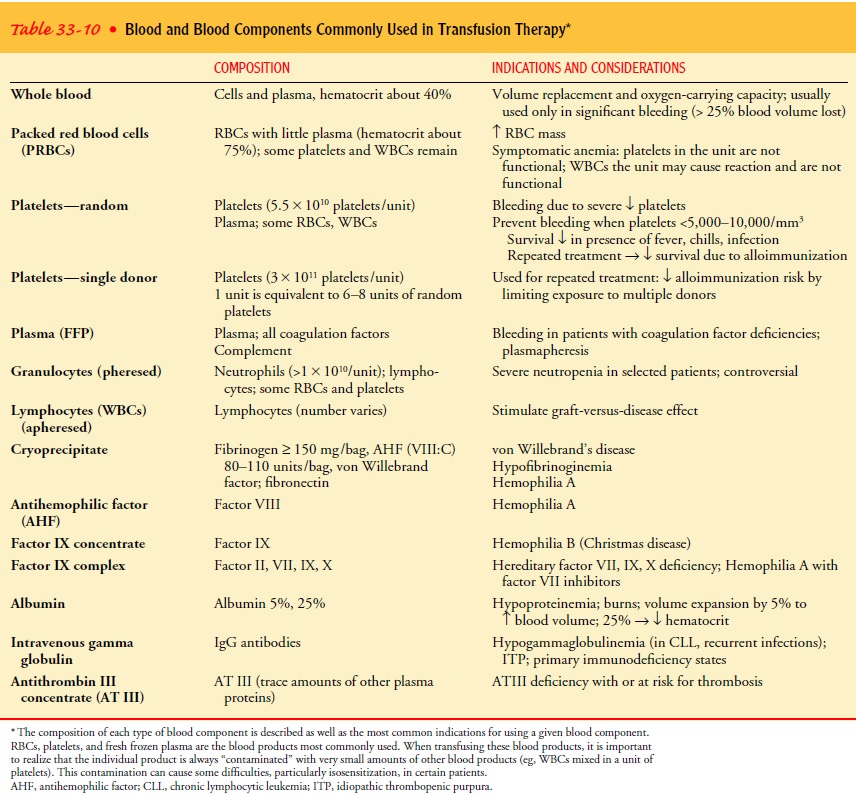
BLOOD AND BLOOD COMPONENT THERAPY
A
single unit of whole blood contains 450 mL of blood and 50 mL of an
anticoagulant. A unit of whole blood can be processed and dispensed for
administration. However, it is more appropriate, economical, and practical to
separate that unit of whole blood into its primary components: RBCs, platelets,
and plasma (WBCs are rarely used; see later discussion). Because the plasma is
re-moved, a unit of RBCs (packed RBCs, PRBCs) is very concen-trated
(hematocrit, approximately 70%). Each component must be processed and stored
differently to maximize the longevity of the viable cells and factors within
it; each individual blood com-ponent has a different storage life. PRBCs are
stored at 4°C. With special
preservatives, they can be stored safely for up to 42 days before they must be
discarded. In contrast, platelets must be stored at room temperature because
they cannot withstand cold temperatures, and they last for only 5 days before
they must be discarded. To prevent clumping, platelets are gently agitated
while stored. Plasma is immediately frozen to maintain the ac-tivity of the
clotting factors within; it lasts for 1 year if it remains frozen. Plasma can
be further pooled and processed into blood derivatives, such as albumin, immune
globulin, factor VIII, and factor IX. Table 33-10 describes each blood
component and how it is commonly used.
SPECIAL PREPARATIONS
Factor VIII concentrate (antihemophilic factor) is a lyophilized, freeze-dried concentrate of pooled fractionated human plasma. It is used in treating hemophilia A. Factor IX concentrate (pro-thrombin complex) is similarly prepared and contains factors II, VII, IX, and X. It is used primarily for treatment of factor IX de-ficiency (hemophilia B). Factor IX concentrate is also useful in treating congenital factor VII and factor X deficiencies.
Plasma
albumin is a large protein molecule that usually stays within vessels and is a
major contributor to plasma oncotic pressure. This protein is used to expand
the blood volume of patients in hypovolemic shock and, rarely, to increase the
concentration of circulating albumin in patients with hypo-albuminemia.
Immune
globulin is a concentrated solution of the antibody IgG; it contains very
little IgA or IgM. It is prepared from large pools of plasma. The intravenous
form (IVIG) is used in various clinical situations to replace inadequate
amounts of IgG in patients who are at risk for recurrent bacterial infection
(eg, those with CLL, those receiving BMT or PBSCT). IVIG, in contrast to all
other fractions of human blood, cells, or plasma, are able to survive being
subjected to heating at 60°C (140°F) for 10 hours to free them of viral contaminants.
Related Topics