Chapter: Medical Surgical Nursing: Assessment and Management of Patients With Hematologic Disorders
Multiple Myeloma
MULTIPLE
MYELOMA
Multiple
myeloma is a malignant disease of the most mature form of B lymphocyte, the
plasma cell. It is not classified as a lymphoma. Plasma cells secrete
immunoglobulins, proteins necessary for anti-body production to fight
infection.
Pathophysiology
In myeloma, the malignant plasma cells
produce an increased amount of a specific immunoglobulin that is nonfunctional.
Func-tional types of immunoglobulin are still produced by nonmalignant plasma
cells, but in lower-than-normal quantity. The specific im-munoglobulin secreted
by the myeloma cells is detectable in the blood or urine and is referred to as
the monoclonal protein, or M protein. This protein serves as a useful marker to
monitor the ex-tent of disease and the patient’s response to therapy. It is
measured by serum or urine protein electrophoresis. Moreover, the patient’s
total protein level is typically elevated, again due to the production of M
protein. Malignant plasma cells also secrete certain substances to stimulate
the creation of new blood vessels to enhance the growth of these clusters of
plasma cells; this process is referred to as angio-genesis. Occasionally the
plasma cells infiltrate other tissue, in which case they are referred to as
plasmacytomas. Plasmacytomas can occur in the sinuses, spinal cord, and soft
tissues. Median sur-vival time is 3 to 5 years. Death usually results from
infection.
Clinical Manifestations
The
classic presenting symptom of multiple myeloma is bone pain, usually in the
back or ribs. Bone pain is reported by two thirds of all patients at diagnosis.
Unlike arthritic pain, the bone pain associated with myeloma increases with
movement and de-creases with rest; patients may report that they have less pain
on awakening but the pain intensity increases during the day. In myeloma, a
substance secreted by the plasma cells, osteoclast ac-tivating factor, as well
as other substances (eg, interleukin-6 [IL-6]) are involved in stimulating
osteoclasts. Both mechanisms appear to be involved in the process of bone
breakdown. Thus, lytic lesions as well as osteoporosis may be seen on bone
x-rays. (They are not well visualized on bone scans.) The bone destruc-tion can
be severe enough to cause fractures, including spinal fractures, which can
impinge on the spinal cord and result in spinal cord compression. It is this
bone destruction that causes significant pain.
If the
bone destruction is fairly extensive, excessive ionized cal-cium is lost from
the bone and enters the serum; patients may there-fore become hypercalcemic
(frequently manifested by excessive thirst, dehydration, constipation, altered
mental status, confusion, and perhaps coma). Renal failure may also be seen;
the configura-tion of the circulating immunoglobulin molecule (particularly the
shape of lambda light chains) can damage the renal tubules.As more and more
malignant plasma cells are produced, the marrow has less space for RBC
production, and the patient can become anemic. This anemia is also caused to a
great extent by a diminished production of erythropoietin (a glycoprotein
necessary for RBC production) by the kidney. Patients may complain of fatigue
and weakness due to the anemia. In the late stage of the disease, a reduced
number of WBCs and platelets may also be seen because the bone marrow is
infiltrated by malignant plasma cells.
When
plasma cells secrete excessive amounts of immunoglob-ulin, particularly IgA, the
serum viscosity can be elevated. Hyper-viscosity may be manifested by bleeding
from the nose or mouth, headache, blurred vision, paresthesias, or heart
failure.
Assessment and Diagnostic Findings
Finding
an elevated monoclonal protein spike in the serum (via serum protein
electrophoresis) or urine (via urine protein elec-trophoresis) or light chain
in the urine (sometimes referred to as Bence Jones protein) is considered to be
a major criterion in the diagnosis of multiple myeloma. The presence of lytic
bone lesions on x-ray aids in the diagnosis, as does the presence of anemia or
hypercalcemia. The diagnosis of myeloma can be confirmed by bone marrow biopsy;
the presence of sheets of plasma cells is the hallmark diagnostic criterion.
Because the infiltration of the mar-row by these malignant plasma cells is not
uniform, the extent of plasma cells may not be increased in a given sample (a
false-neg-ative result).
Gerontologic Considerations
The
incidence of multiple myeloma increases with age; the disease rarely occurs in
patients younger than 40 years of age. Because of the increasing older
population, more patients are seeking treat-ment for this disease. BMT or PBSCT
is an option that can pro-long remission and potentially cure some patients.
However, it is unavailable to most because of age limitations. Back pain, which
is often a presenting symptom in this disease, should be closely in-vestigated
in elderly patients.
Medical Management
There
is no cure for multiple myeloma. Even BMT or PBSCT is considered by most
authorities to extend remission rather than pro-vide a cure. However, for many
patients, it is possible to control the illness and maintain their level of
functioning quite well for several years or longer. Chemotherapy is the primary
treatment; cortico-steroids, particularly dexamethasone (Decadron), are
especially effective and are often combined with other agents (such as
melphalan (Alkeran), cyclophosphamide (Cytoxan), doxorubicin (Adriamyein),
vincristine (Oncouin), and BCNU (Carmustine).
Radiation
therapy is very useful in strengthening a specific bone lesion, particularly
one at risk for bone fracture or spinal cord compression. It is also useful in
relieving bone pain and reducing the size of plasma cell tumors that occur
outside the skeletal sys-tem. However, because it is a nonsystemic form of
treatment, it does not diminish the source of the bone problems (ie, the
pro-duction of malignant plasma cells). Therefore, radiation therapy is
typically used with systemic treatment such as chemotherapy.
The
biologic agent alpha-interferon has been used successfully to maintain
remission in selected types of myeloma, particularly IgA type; however, its
role in prolonging survival is controversial. Newer forms of bisphosphonates,
such as pamidronate (Aredia) and zoledronic acid (Zometa), have been shown to
strengthen bone in this disease (by diminishing the secretion of osteoclast
activating factor) (Terpos et al., 2000), controlling bone pain and potentially
preventing bone fracture. They are also effective in managing and preventing
hypercalcemia. Some evidence suggests that bisphosphonates may actually have
activity against the mye-loma cells themselves by inhibiting a growth factor
necessary for myeloma cell survival (Berenson, 2001) (see later discussion).
When
patients manifest signs and symptoms of hyperviscosity, plasmapheresis may be
used to lower the immunoglobulin level. Symptoms may be more useful than serum
viscosity levels in de-termining the need for this intervention.
Recent
advances in the understanding of the process of angio-genesis have resulted in
new therapeutic options. The sedative thalidomide (Thalomid), initially used as
an antiemetic, has sig-nificant antimyeloma effects. It inhibits cytokines
necessary for new vascular generation, such as, vascular endothelial growth
fac-tor (VEGF) and for myeloma cell growth and survival, such as IL-6 and tumor
necrosis factor), by boosting the body’s immune response against the tumor and
by creating favorable conditions for apoptosis of the myeloma cells.
Thalidomide is effective in re-fractory myeloma and in “smoldering” disease
states, and may pre-vent progression to a more active state. Thalidomide is not
a typical chemotherapeutic agent and has a unique side effect pro-file. Fatigue,
dizziness, constipation, rash, and peripheral neu-ropathy are commonly
encountered; myelosuppression is not (Goldman, 2001). Thalidomide is
contraindicated in pregnancy because of associated severe birth defects.
Nursing Management
Pain
management is very important in this patient population. NSAIDs can be very
useful for mild pain, or in combination with opioid analgesics. However, care
needs to be taken, because NSAIDs can cause renal dysfunction. Patients need to
be educated about activity restrictions (eg, lifting no more than 10 pounds,
use of proper body mechanics). Braces are occasionally needed to pro-vide
support to the spinal column.
Patients
also need to be instructed about the signs and symp-toms of hypercalcemia.
Maintaining mobility and hydration is important to diminish exacerbations of
this complication; how-ever, the primary cause is the disease itself. Renal
function should also be monitored closely. Renal failure can become severe, and
dialysis may be needed. Maintaining high urine output (3 L/day) can be very
useful in preventing this complication.
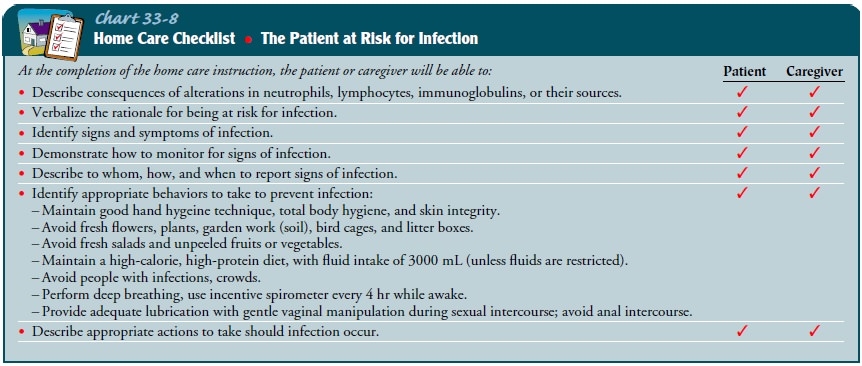
Because
antibody production is impaired, infections, particu-larly bacterial
infections, are common and can be life-threatening. Patients need to be
instructed in appropriate infection prevention measures (see Chart 33-8) and
should be advised to contact their health care provider immediately if they
have a fever or other signs and symptoms of infection. Patients should receive
Pneumovax and flu vaccines. Prophylactic antibiotics are sometimes used.
In-travenous gamma globulin (IVIG) can be useful for patients with recurrent
infections.
Related Topics