with Answers, Solution | Atomic and Nuclear Physics | Physics - Numerical Exercises Problems | 12th Physics : UNIT 9 : Atomic and Nuclear Physics
Chapter: 12th Physics : UNIT 9 : Atomic and Nuclear Physics
Numerical Exercises Problems
Atomic and Nuclear Physics
Exercises
1. Consider two hydrogen atoms HA and HB in ground state. Assume that hydrogen atom HA is at rest and hydrogen atom HB is moving with a speed and make head-on collide on the stationary hydrogen atom HA. After the strike, both of them move together. What is minimum value of the kinetic energy of the moving hydrogen atom HB, such that any one of the hydrogen atoms reaches one of the excitation state.
[Ans: 20.4 eV]
Solution:

2. In the Bohr atom model, the frequency of transitions is given by the following expression
v = Rc ( 1/n2 – 1/m2 ) , where n < m,
Consider the following transitions:
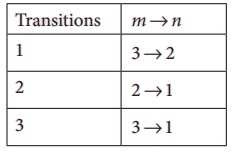
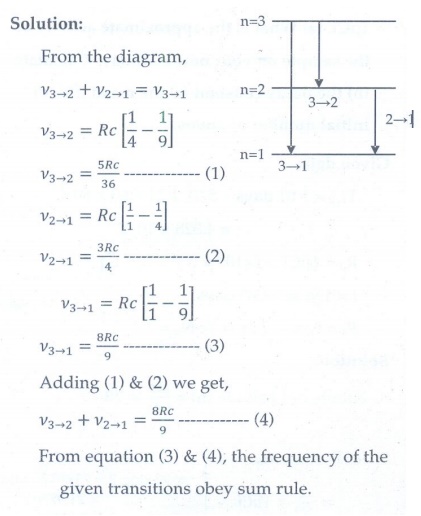
Show that the frequency of these transitions obey sum rule (which is known as Ritz combination principle)
[Ans: v3→2 + v2→1 = v3→1]
3. (a) A hydrogen atom is excited by radiation of wavelength 97.5 nm. Find the principal quantum number of the excited state.
(b) Show that the total number of lines in emission spectrum is n(n-1) / 2 and compute the total number of possible lines in emission spectrum.
[Ans: (a) n = 4 (b) 6 possible transitions]

4. Calculate the radius of the earth if the density of the earth is equal to the density of the nucleus.[mass of earth 5.97 × 1024 kg ].
[Ans:180m]
Given data:
Mass
of the earth = 5.97 x 1024 kg
Density
of earth = Density of nucleus
=
2.3 x 1017 kg m-3
Solution:
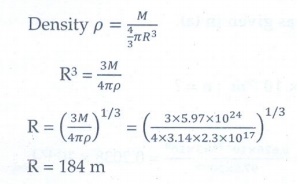
5. Calculate the mass defect and the binding energy per nucleon of the 10847Ag nucleus. [atomic mass of Ag = 107.905949]
Ans: [∆m = 0.990391u and B.E = 8.5MeV / A ]
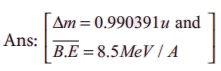
Given data:
M
= 107.905949 u
Z
= 47 mH = 1.007825u
N
= 61 mn =1.008665
u
Solution:

6. Half lives of two radioactive elements A and B are 20 minutes and 40 minutes respectively. Initially, the samples have equal number of nuclei. Calculate the ratio of decayed numbers of A and B nuclei after 80 minutes.
[Ans: 5:4]
Given data:
T1/2 of A = 20
min
T1/2 of B =
40 min ; Total time = 80 min
NOA = NOB
NOA - NA / NOB - NB = ?
Solutions:
Decayed
number of nuclei = No – N
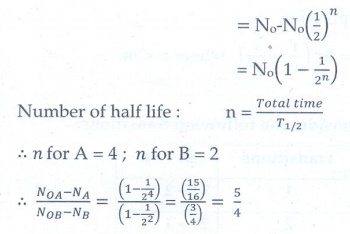
The
ratio of decayed numbers of A and B is 5 : 4.
7. On your birthday, you measure the activity of the sample 210Bi which has a half-life of 5.01 days. The initial activity that you measure is 1μCi . (a) What is the approximate activity of the sample on your next birthday? Calculate (b) the decay constant (c) the mean life (d) initial number of atoms.
[Ans: (a) 10-22 μCi (b) 1.6 ×10-6 s-1 (c) 7.24days (d) 2.31×1010 ]
Given data:
T1/2
= 5.01 days = 5.01 × 24 × 60 × 60 s
= 4.328 × 105 s
R0
= 1 μCi = 1 × 10-6 × 3.7 × 1010 Bq
t = 1 year = 365 days
Rt
= ? ; λ=?; τ = ? ; N0 = ?
Solution:

8. Calculate the time required for 60% of a sample of radon undergo decay. Given T1/2 of radon =3.8 days
[Ans: 5.022 days]
Given data:
T1/2 = 3.8
days; λ= 0.693/3.8 per day
Amount of
sample decayed = 60% of No
Amount of
sample present (N) = 40% of No
No
is initial amount of sample, t = ?
Solution:
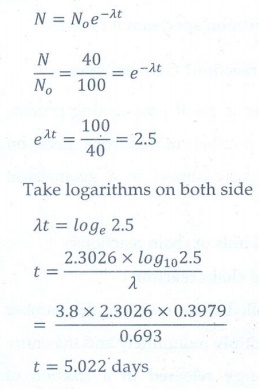
9. Assuming that energy released by the fission of a single 23592U nucleus is 200MeV, calculate the number of fissions per second required to produce 1 watt power.
[Ans: 3.125 ×1010 ]
Given data:
Energy
released during fission of single
23492U =
200MeV
Power
= 1 watt = 1 J/s
No.
of fissions per second required to produce
1W
power = ?
Solution:
Energy
produced = no. of fissions per second × energy released per fission
No.of
fissions per second = energy produced / energy released per fission
=
1 / ( 200 ×106 ×1.6 ×10-19)
=
1/ (320 × 10-13)
=
1013/320 = 3.125 ×1010
No.
of fissions per second = 3.125 × 1010
10. Show that the mass of radium ( 22688Ra ) with an activity of 1 curie is almost a gram. Given T1/2 =1600 years.
Given: T1/2 =1600
years (PTA-6)
Given data:
Ro
= 1Ci= 3.7 × 1010 Bq
T1/2 = 1600 years
Mass
of radium with an activity of 1 Ci= ?
Solution:

∴ The mass of radium with an activity of 1 Ci
is almost a gram.
11. Characol pieces of tree is found from an archeological site. The carbon-14 content of this characol is only 17.5% that of equivalent sample of carbon from a living tree. What is the age of tree?
[Ans: 1.44 × 104 yr]
Given data:
The
amount of 14C present (N) = 17.5% of No
T1/2 = 5730
years
Age
of the tree (t) = ?
Solution:
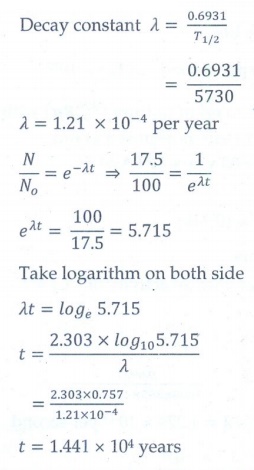
Related Topics