Chapter: 12th Physics : UNIT 9 : Atomic and Nuclear Physics
Law of radioactive decay
Law of radioactive decay
In the previous section, the decay
process of a single radioactive nucleus was discussed. In practice, we have
bulk material of radioactive sample which contains a vast number of the radioactive
nuclei and not all the radioactive nucleus in a sample decay at the same time.
It decays over a period of time and this decay is basically a random process.
It implies that we cannot predict which nucleus is going to decay or rather we
can determine like probabilistic basis (like tossing a coin). We can calculate
approximately how many nuclei in a sample are decayed over a period of time.
At any instant t, the number of decays per unit time, called rate of decay (dN/dt) is proportional to the number of nuclei ( N ) at the same instant.
dN/dt ∝ N
By introducing a proportionality constant,
the relation can be written as

Here proportionality constant λ is
called decay constant which is different for different radioactive sample and
the negative sign in the equation implies that the N is decreasing with time.
By rewriting the equation (8.32), we
get
dN = −λNdt (8.33)

Here dN represents the number of nuclei decaying in the time interval dt.
Let us assume that at time t = 0
s, the number of nuclei present in the radioactive sample is N0. By integrating the
equation (8.33), we can calculate the number of undecayed nuclei N at any time t.
From equation (8.33), we get
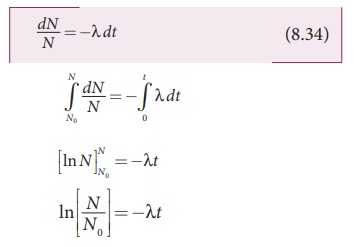
Taking exponentials on both sides,
we get

N = N0e-λt (8.35)
[Note: elnx = ey
⇒ x = e y ]
Equation (8.35) is called the law of
radioactive decay. Here N denotes the
number of undecayed nuclei present at any time t and N0 denotes
the number of nuclei at initial time t=0.
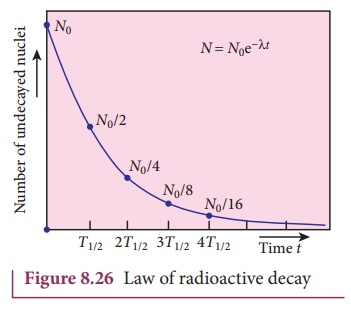
Note that the number of atoms is decreasing exponentially over the time. This
implies that the time taken for all the radioactive nuclei to decay will be
infinite. Equation (8.35) is plotted in Figure 8.26.
We can also define another useful
quantity called activity (R) or decay rate which is the number of nuclei
decayed per second and it is denoted as R
= |dN/dt|. Note that activity R is a positive quantity.
From equation (8.35), we get
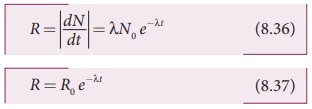
where R0 = λN0
The equation (8.37) is also
equivalent to radioactive law of decay. Here R0 is the activity of the sample at t=0 and R is the activity of the sample at any
time t. From equation (8.37),
activity also shows exponential decay behavior. The activity R also can be expressed in terms of
number of undecayed atoms present at any time t.
From equation (8.37), since N = N0e−λt , we write
R = λN (8.38)

Equation (8.35) implies that the
activity at any time t is equal to
the product of decay constant and number of undecayed nuclei at the same time t. Since N decreases over time, R also
decreases.
The SI unit of activity R is Becquerel and one Becquerel (Bq) is
equal to one decay per second. There is also another standard unit for the
activity called Curie(Ci).
1 Curie =1 Ci = 3.7 ×1010
decays per second
1 Ci = 3.7 ×1010 Bq
Initially one curie was defined as
number of decays per second in 1 g of radium and it is equal to 3.7 ×1010
decays/s.
Related Topics