Chapter: Biochemistry: Lipids
Structure of triacyl glycerol
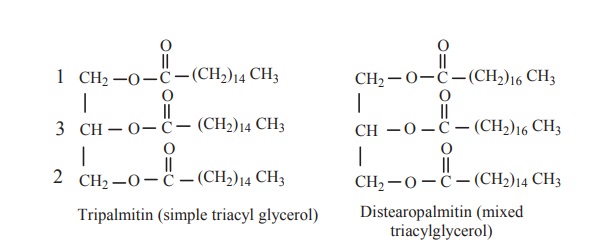
Structure of triacyl glycerol
Triacylglycerols are simple lipids in which
glycerol backbone is esterified with three fatty acids. This form the major
part of dietary lipids. They are stored in adipose tissue and serve as
concentrated fuel reserve of the body.
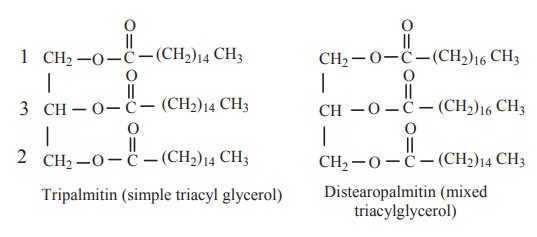
If the three hydroxyl groups are esterified
with same type of fatty acid then the lipid is called as simple glyceride. If
the three hydroxyl groups are esterified with different type of fatty acids,
the lipid is called as mixed glyceride.
1. Physical Properties
Physical
·
Triacylglycerols
are non polar, hydrophobic molecules, insoluble in water, but soluble in
organic solvents.
·
Specific
gravity of fats is lower than water. Therefore fats and oils float on water.
·
Melting
point of triacylglycerol is related to the chain length and degree of
unsaturation of fatty acids. The longer the chain length, the higher the
melting point and greater the number of double bonds, the lower the melting
point.
·
They are
tasteless, odourless, colourless and neutral in solution.
·
They are
themselves good solvents for other fats.
2. Chemical properties
1. Hydrolysis :
On boiling with water at 200°C,
triacyl glycerols are hydrolysed to glycerol and fatty acids in a stepwise
manner.
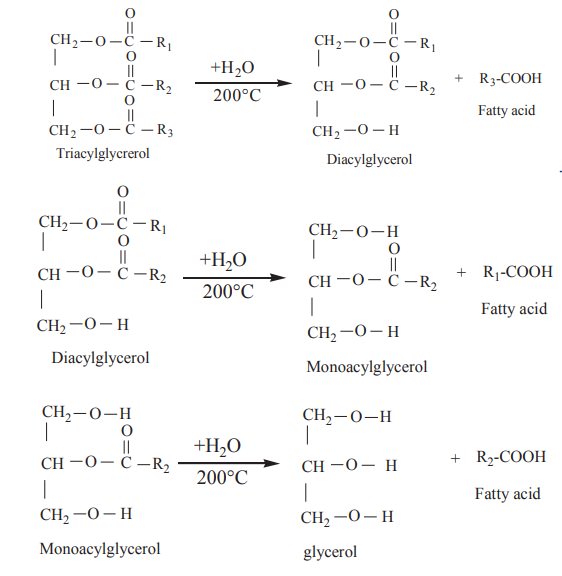
The reaction can also be catalysed by the
enzymes lipases.
2. Hydrogenation
Hydrogenation of unsaturated fatty acids
present in the fats, lead to the formation of saturated fats. Hydrogenation
elevates the melting point. Thus an oil is converted to a solid fat.
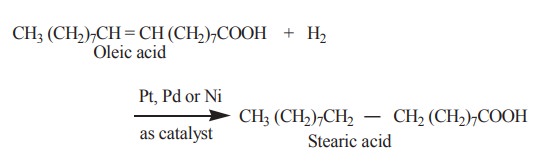
This reaction is of great commercial importance
since it permits transformation of inexpensive and unsaturated liquid vegetable
fats into solid fats. The latter are used in the manufacture of candles,
vegetable shortenings like vanaspathi and oleomargarine.
3. Saponification
Boiling with an alcoholic solution of strong
metallic alkali hydrolyses triacyl glycerol into soap and fatty acid. This
process is called as saponification.
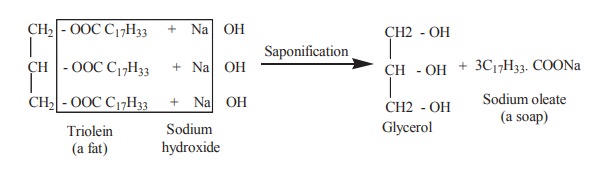
Soaps are important cleansing agents. Their
cleansing property is due to their emulsifying action. This is accomphlised by
means of negative charge the soap anion confers on oil droplets. The
electrostatic repulsion then prevents the coalescene of soap and results in the
removal of dirt particles.
4. Halogenation
Unsaturated fatty acids in the triacyl glycerol
take up chlorine, bromine and iodine atoms at their double bonds to form
saturated halogenated derivatives.
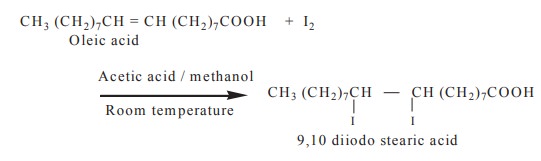
5. Rancidity
On storage, unsaturated fatty acids present in
the fat are likely to undergo oxidation and hydrolytic cleavage in the presence
of lipases present in the fat itself or secreted by the contaminating
microorganisms. This leads to change of colour and odour of the fat. This
change is called as rancidity. This occurs due to the formation of peroxides at
the double bonds of unsaturated fatty acids. Rancidity can be prevented by
certain antioxidants such as vitamin -E, gallic acid, butylated hydroxy toluene
etc.Vegetable oils are less rancid because of the presence of natural
antioxidants such as vitamin E and carotenoids.
3. Quantitative tests
There are certain chemical constants used for
the characterisation of fats.
1. Acid Number
It is the number of milligrams of KOH required
to neutralize the free fatty acids present in 1 gm of fat. The acid number,
thus, tells us about the quantity of free fatty acid present in a fat.
Obviously, a fat which has been processed and stored properly has a very low
acid number.
2. Saponification number
It is the number of milligrams of KOH requried
to saponify 1 gm of fat. The saponification number, thus, provides information
on the average chain length of the fatty acids in the fat. The saponification
number varies inversely with the chain length of the fatty acids. The shorter
the average chain length of the fatty acids, the higher is the saponification
number.
3. Iodine number
It is the number of grams of iodine absorbed by
100 gm of fat. The iodine number is, thus, a measure of the degree of
unsaturation of the fatty acids in the fat. The iodine number gives no
indication as to the number of double bonds present in the fatty acid
molecules.
4. Polenske number
It is the number of millilitres of 0.1 N KOH
required to neutralize the insoluble fatty acids. This indicates the level of
non volatile fatty acids present in the fat.
5. Reichert - Meissl number
It is the number of millilitres of 0.1 N KOH
required to neutralize the soluble, volatile fatty acids derived from 5 gm of
fat. The Reichert- Meissl number thus, measures the quantity of short chain
fatty acids in the fat molecule.
4. Acetyl number
It is the number of milligrams of KOH required
to neutralize the acetic acid obtained by saponification of 1 gm of fat after
it has been acetylated. The acetyl number is, thus, a measure of the number of
OH groups in the fat.
Related Topics