Chapter: Plant Biochemistry: Lipids are membrane constituents and function as carbon stores
Triacylglycerols are synthesized in the membranes of the endoplasmatic reticulum
Triacylglycerols are synthesized in the membranes of the endoplasmatic reticulum
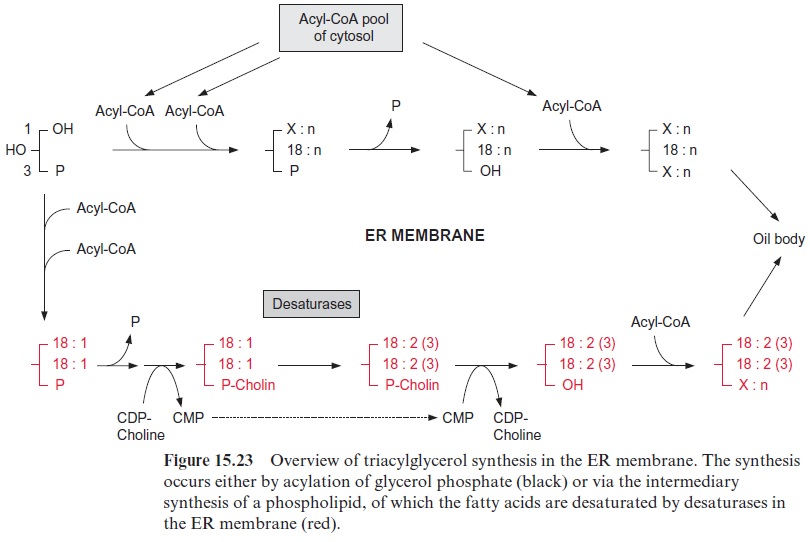
The lipid content of mature seeds can amount to 45% of the dry weight, but in some cases (e.g., Ricinus) up to 85%. The precursor for the synthesis of tri-acylglycerols is again glycerol 3-phosphate. There are at least four pathways for its synthesis, the two most important will be discussed here (Fig. 15.23):
Phosphatidic acid is formed by acylation of the hydroxyl groups at posi-tion C-1 and C-2 of glycerol and, after hydrolysis of the phosphate resi-due, the C-3-position is also acylated. The total cytosolic acyl CoA pool is available for these acylations, but due to the eukaryotic pathway, posi-tion 2 is mostly esterified by a C18 acyl residue (Fig. 15.21).
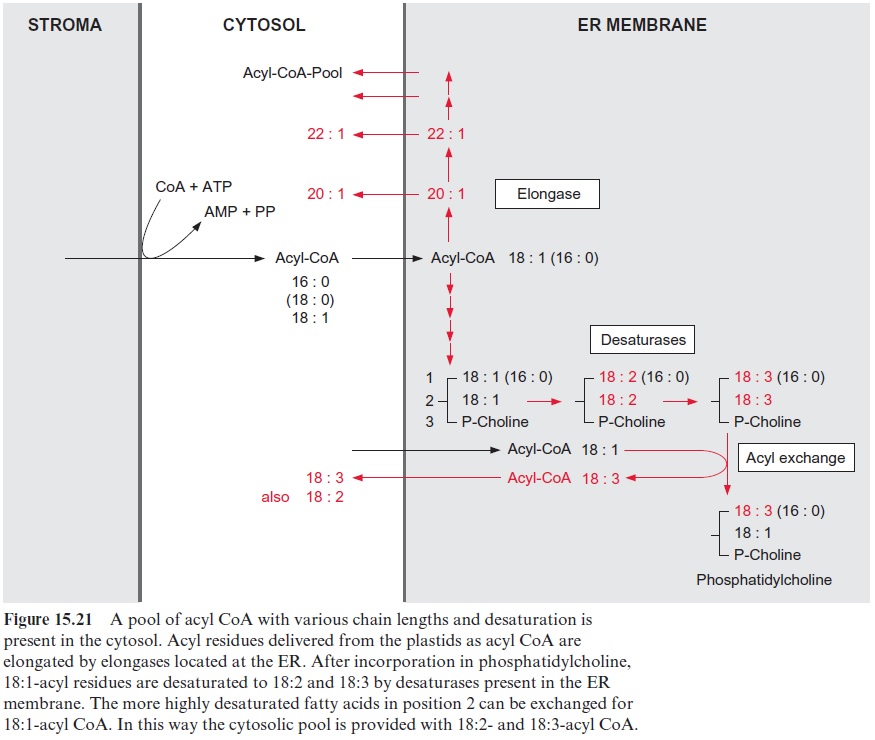
Phosphatidylcholine is formed first, and its acyl residues are further desaturated. The choline phosphate residue is transferred to CMP, and the corresponding diacylglycerol is esterified with a third acyl residue.
This pathway operates frequently during the synthesis of highly unsatu-rated triacylglycerols.
Cross-connections exist between the two pathways, but for the sake of simplicity these are not discussed in Figure 15.23.
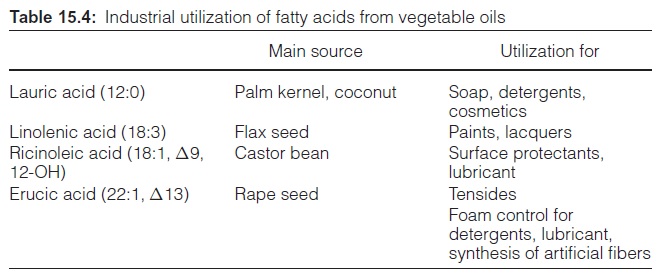
Plant fat is used for human nutrition and also as a raw material in industry
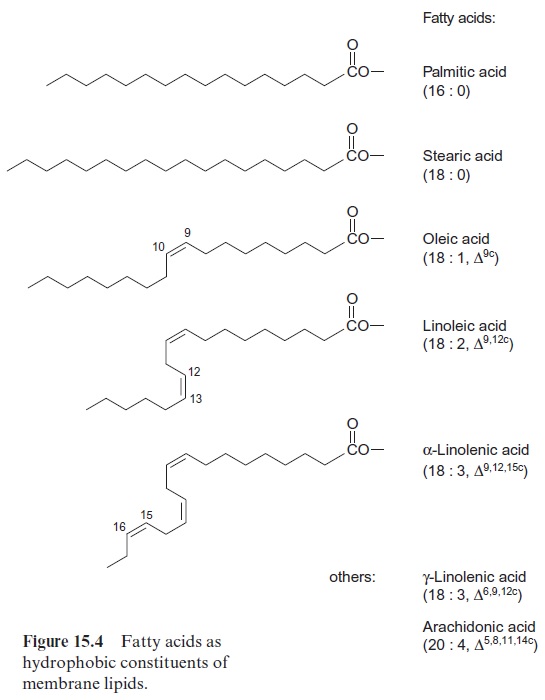
About 20% of the human caloric nutritional uptake in industrialized countries is due to the consumption of plant fats. Plant fats have a nutritional advan-tage over animal fats since they contain a higher portion of unsaturated fatty acids. Human metabolism requires unsaturated fatty acids with two or more carbon-carbon double bonds, but is not able to introduce double bonds at position 12 and 15. This is why linoleic acid and linolenic acid (Fig. 15.4) are essential fatty acids, which are absolutely necessary in the human diet.
Plant fats are also important as industrial raw materials. Fatty acids are obtained after the hydrolysis of triacylglycerols. They have been in use as alkali salts in soap since ancient times. Fatty acid alcohols and fatty acid methyl esters are also used as detergents. Moreover, fatty acid methyl esters synthesized from rape seed oil are used as car fuel (bio diesel). The high quantities of glycerol released during the hydrolysis of fats are an impor-tant industrial raw material.
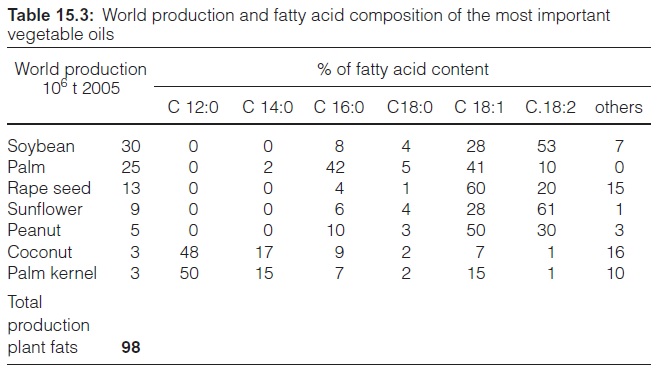
Table 15.3 shows that the fatty acid composition of various plant oils is very divergent. Triacylglycerols of some plants comprise large quantities of rare fatty acids, which are used for industrial purposes (Table 15.4). Oil from palm kernels has a high content of the short-chain lauric acid (12:0), which is used for the production of detergents and cosmetics. Large planta-tions of oil palms in Southeast Asia ensure its supply to the oleochemical industry. Linolenic acid from European flax seed is used for the production of paints. Ricinoleic acid, a rare fatty acid that comprises a hydroxyl group at the C-12-position, accounts for about 90% of the fatty acids in castor oil. It is used in industry as a lubricant and as a surface protectant. Erucic acid is used for similar purposes. Earlier varieties of rape seed had erucic acid in their triacylglycerols, and for this reason rape seed oil was then of inferior value for human nutrition. About 40 years ago, successful crossings led to a breakthrough in the breeding of rape seed free of erucic acid, with the result that rape seed oil is now highly suitable for human consumption. The values given in Table 15.3 are indicative for this new type of rape seed, which is now cultivated worldwide. However, rape seed varieties containing a high per-centage of erucic acid are recently cultivated again for industrial demands.
Plant fats are customized by genetic engineering
The progress in gene technology now makes it possible to alter the quality of plant fats in a defined way by changing the enzymatic profile of the cell. The procedures for the introduction of a new enzyme into a cell, or for eliminating the activity of an enzyme present in the cell. Three cases of the alteration of oil crops by genetic engineering will be illustrated here.
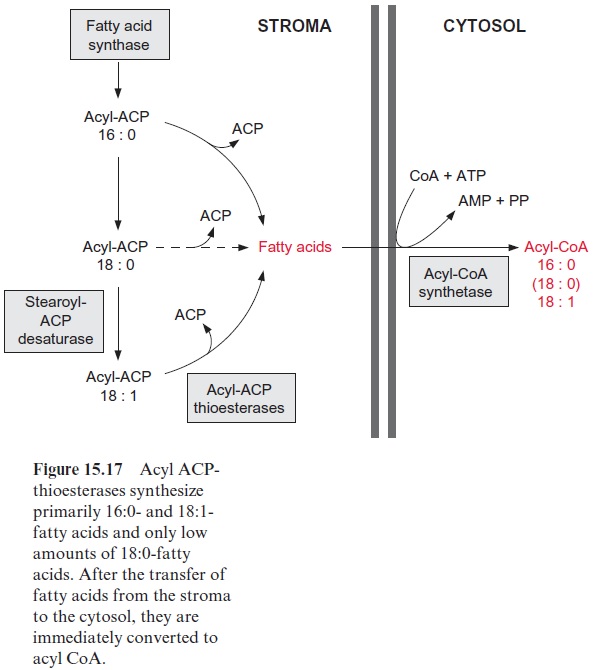
1. The lauric acid present in palm kernel and coconut oil (12:0) is an impor-tant raw material for the production of soaps, detergents, and cosmetics. Recently, rape seed plants that contained oil with a lauric acid content of 66% were generated by genetic transformation. The synthesis of fatty acids is terminated by the hydrolysis of acyl ACP (Fig. 15.17). An acyl ACP thioesterase, which specifically hydrolyzes lauroyl ACP, was isolated and cloned from the seeds of the California bay tree (Umbellularia califor-nica), which contains a very high proportion of lauric acid in its storage lipids. The introduction of the gene for this acyl ACP thioesterase into the developing rape seed terminates its fatty acid synthesis at acyl (12:0), and the released lauric acid is incorporated into the seed oil. Field studies have shown that these plants grow normally and yields are as expected.
2. A relatively high content of stearic acid (18:0) improves the heat stability of fats for deep frying and for the production of margarine. The stearic acid content in rape seed oil has been increased from 1%–2% to 40% by decreasing the activity of stearoyl ACP desaturase using antisense technique. On the other hand, genetic engineering has been employed to increase highly unsaturated fatty acids (e.g., in rape seed) providing the oil with a higher nutritional quality. In soybeans, the pro-portion of double unsaturated fatty acids in the oil could be increased to 30%. Nutritional physiologists discovered that highly unsaturated fatty acids (20:4, 20:5, 22:6), which are only present in fish oil, are very benefi-cial for human health. Investigations by genetic engineering are now in progress to develop rape lines with highly unsaturated fatty acids of C20 and C22 length. This is an example where gene technology is applied for the production of “healthy food.”
Erucic acid (Fig. 15.24) could be an important industrial raw material, for instance for the synthesis of synthetic fibers and for foam control in detergents. Presently, however, its utilization is limited, since conventional breeding has not succeeded in increasing the erucic acid content to more than 50% of the fatty acids in the seed oil. The costs of separating erucic acid and disposal of the other fatty acids are so high that for many purposes the industrial use of rape seed oil as a source of erucic acid from presently available cultivars is not economically viable. Attempts are being made to increase further the erucic acid content of rape seed oil by overexpression of elongase genes and by transferring genes encoding enzymes that catalyze the specific incorporation of erucic acid to the C-2-position of triacylglycerols. If this were to be successful, present petrochemical-based industrial processes could be replaced with processes using rape seed oil as a renewable resource.
Related Topics