Chapter: Plant Biochemistry: Lipids are membrane constituents and function as carbon stores
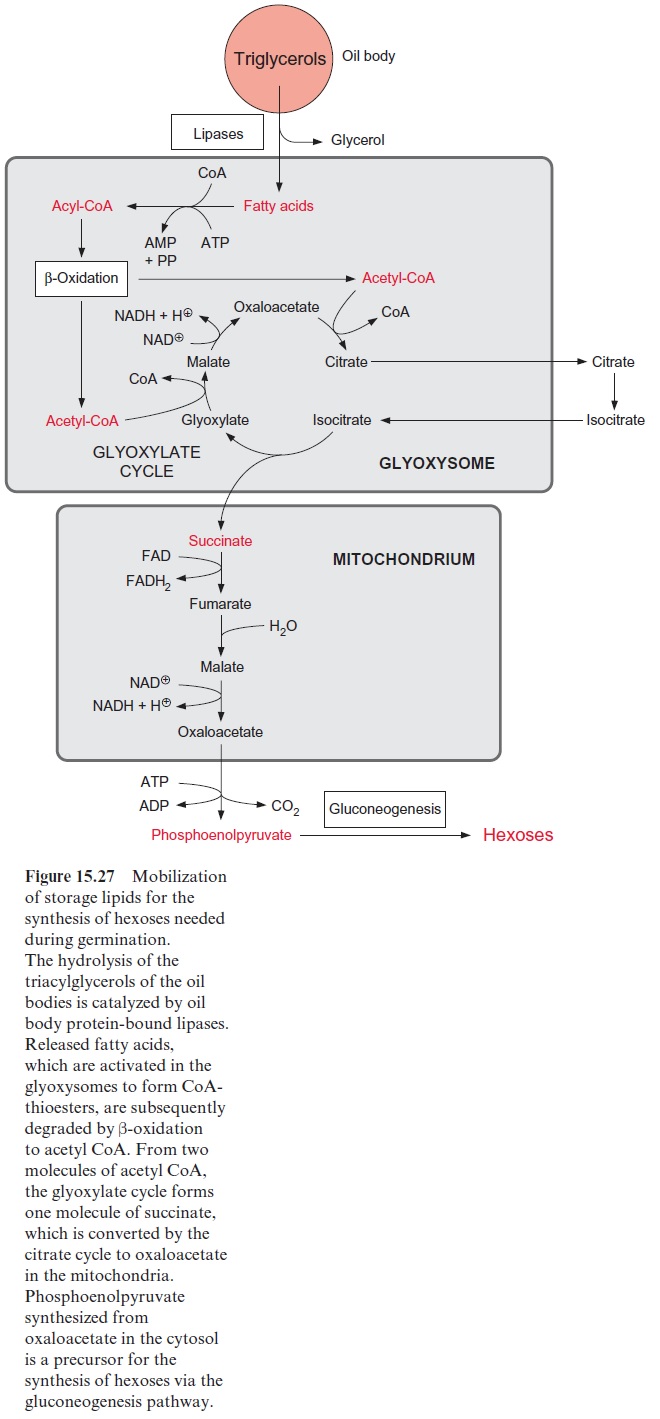
Storage lipids are mobilized for the production of carbohydrates in the glyoxysomes during seed germination
Storage lipids are mobilized for the production of carbohydrates in the glyoxysomes during seed germination
At the onset of germination, storage proteins are degraded to amino acids, from which enzymes required, for example, for the mobilization of the storage lipids, are synthesized. These enzymes include lipases, which cat-alyze the hydrolysis of triacylglycerols to glycerol and fatty acids. They bind to the oil body proteins . The glycerolreleased by the hydrolysis of triacylglycerol, after phosphorylation to glycerol 3-phosphate and its subse-quent oxidation to dihydroxyacetone phosphate, is fed into thegluconeogenesis or glycolytic pathway . The free fatty acids are first activated by reaction with CoA to form thioesters and are then degraded to acetyl CoA by β-oxidation (Fig. 15.25). This process proceeds in specialized peroxisomes called glyoxysomes. It has been observed that for a transfer of fatty acids, oil bodies and glyoxysomes come into close contact with each other.
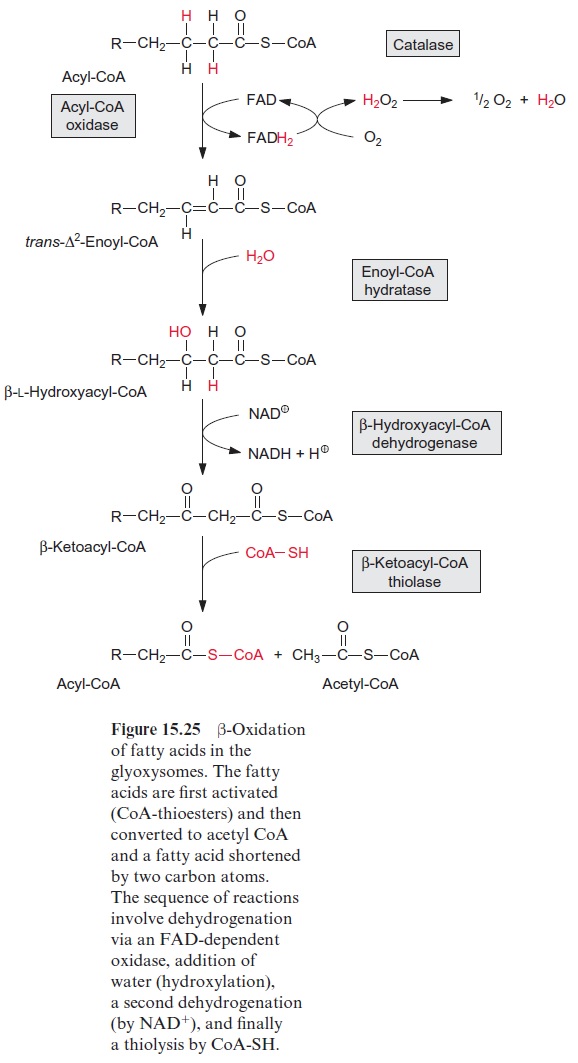
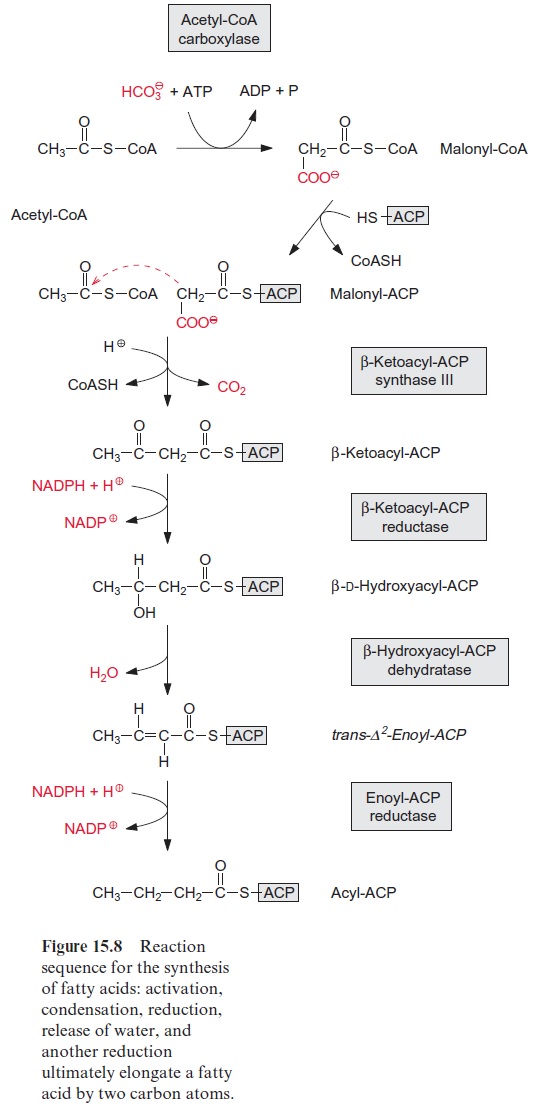
Although in principle β-oxidation represents the reversal of fatty acid synthesis (Fig. 15.8), there are distinct differences that enable high metabolic fluxes through these two metabolic pathways operating in opposite direc-tions. The differences between β-oxidation and fatty acid synthesis are:
1. In acyl CoA dehydrogenation, hydrogen is transferred via an FAD-dependent oxidase to O2 to form H2O2. A catalase irreversibly eliminates the toxic H2O2 at the site of its production by conversion to water and oxygen . Sometimes, for example at a very high rate of - oxidation during seed maturation, the amount of H2O2 can be so high that it leaks out of the glyoxysomes causing cell damage.
2. β-L Hydroxyacyl CoA is synthesized during the hydration of enoyl CoA, in contrast to the corresponding D-enantiomer during fatty acid synthesis.
3. Hydrogen is transferred to NAD+ during the second dehydrogenation step. Normally, most of the NAD + in the cell is highly oxidized , driving the reaction to hydroxyacyl CoA oxidation. It is not known which reactions utilize the NADH synthesized in the peroxisomes.
4. In an irreversible reaction, CoA-SH-mediated thiolysis cleaves β-ketoacyl CoA to synthesize one molecule each of acetyl CoA and of acyl CoA shortened by two C atoms.
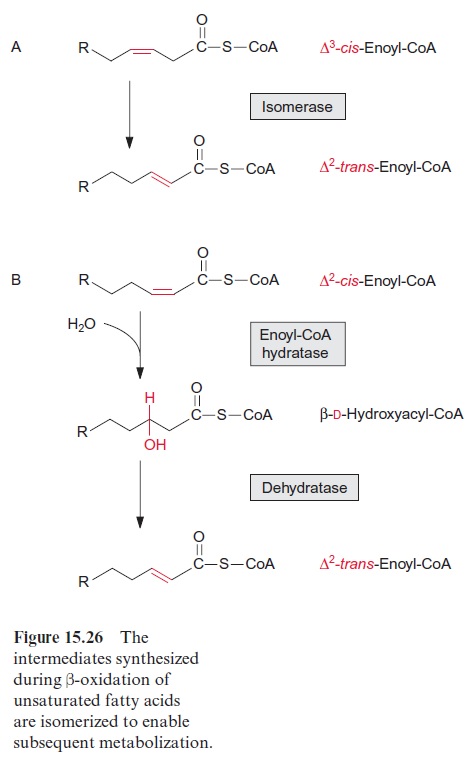
During the degradation of unsaturated fatty acids, sometimes interme-diate products are produced that cannot be metabolized by the reactions of -oxidation. Δ3-cis-enoyl CoA (Fig. 15.26), which is synthesized during the degradation of oleic acid, is converted by an isomerase which shifts the double bond to Δ2-trans-enoyl CoA, an intermediate of β-oxidation. In the β-oxidation of linoleic or linolenic acid, the second double bond in the corres ponding intermediate is at the correct position but in thecis-configuration. Consequently its hydration by enoyl CoA hydratase results in the formation of β-D-hydroxyacyl CoA. The latter is converted by a dehydratase to ∆2-trans-enoyl-CoA, which is an intermediate of -oxidation.
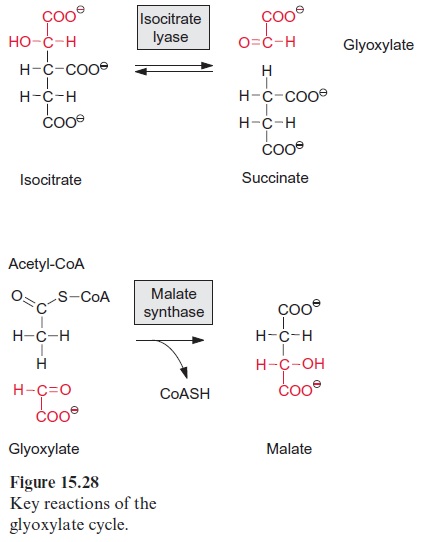
The glyoxylate cycle enables plants to synthesize hexoses from acetyl CoA
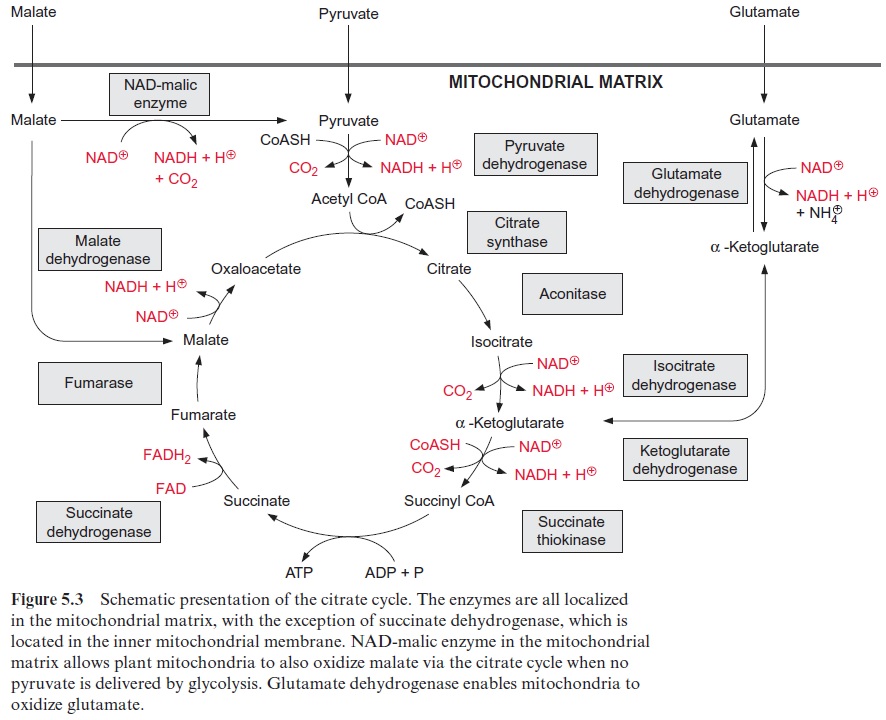
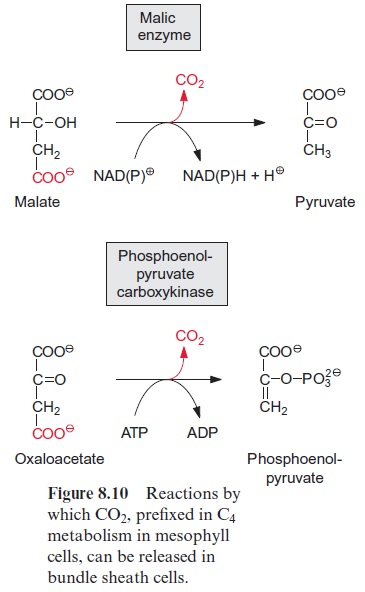
In contrast to animals, which are unable to synthesize glucose from acetyl CoA, plants are capable of gluconeogenesis from lipids, as they possess the enzymes of the glyoxylate cycle (Fig. 15.27). Like β-oxidation, this cycle is located in the glyoxysomes, which are named after this cycle. The two starting reactions of the cycle are identical to those of the citrate cycle (see Fig. 5.3). Acetyl CoA condenses with oxaloacetate, as catalyzed by citrate synthase, to produce citrate, which is then converted by aconitase to isocitrate. The following reaction with isocitrate, however, is a specialty of the glyoxylate cycle: isocitrate is split by isocitrate lyase into succinate and glyoxylate (Fig. 15.28). Malate synthase, the second special enzyme of the glyoxylate cycle, catalyzes the instantaneous condensation of glyoxylate with acetyl CoA to synthesize malate. Due to the hydrolysis of the CoA-thioester this reaction becomes irreversible. As in the citrate cycle, malate is oxidized by malate dehydrogenase to oxaloacetate, thus completing the glyoxylate cycle. In this way one molecule of succinate is generated from two molecules of acetyl CoA. The succinate is transferred to the mitochondria and converted to oxaloacetate by a partial reaction of the citrate cycle. The oxaloacetate is released from the mitochondria by the oxaloacetate translocator and trans-formed in the cytosol by phosphoenolpyruvate carboxykinase to phos-phoenolpyruvate (Fig. 8.10). Phosphoenolpyruvate is a precursor for the synthesis of hexoses by the gluconeogenesis pathway and for other biosyn-thetic processes.
Reactions with toxic intermediates take place in peroxisomes
There is a simple explanation for the fact that β-oxidation and the closely related glyoxylate cycle proceed in specialized peroxisomes (glyoxysomes). As in photorespiration, in which peroxisomes participate , the toxic compounds H2O2 and glyoxylate are also synthesized as intermediates in the conversion of fatty acids to succinate. Compartmentation in the peroxisomes prevents these toxic compounds entering the cytosol . β-Oxidation also takes place, although at a much lower rate, in leaf peroxisomes, where it serves the purpose of recycling the fatty acids that are no longer required or have been damaged. This recycling plays a role in senescence, when carbohydrates are synthesized from the degradation products of membrane lipids in the senescent leaves and are transferred to the stem via the phloem. During senescence, one can indeed observe a differentiation of leaf peroxisomes into glyoxysomes (alternatively termed gerontosomes). The hydrophobic amino acids leucine and valine are also degraded in peroxisomes.
Related Topics