Chapter: Biochemistry: Lipids and Proteins Are Associated in Biological Membranes
The Functions of Biological Membranes
The Functions of Membranes
As
already mentioned, three important functions take place in or on membranes (in
addition to the structural role of membranes as the boundaries and con-tainers
of all cells and of the organelles within eukaryotic cells). The first of these
functions is transport. Membranes are
semipermeable barriers to the flow of substances into and out of cells and
organelles. Transport through the membrane can involve the lipid bilayer as
well as the membrane proteins. The other two
How does transport through membranes take place?
The most important question about transport of
substances across biological membranes is whether the process requires the cell
to expend energy. In passivetransport, a
substance moves from a region of higher concentration to one oflower
concentration. In other words, the movement of the substance is in the same
direction as a concentration gradient,
and the cell does not expend energy. In active
transport, a substance moves from a region of lower concentration to one of
higher concentration (against a concentration gradient), and this process
requires the cell to expend energy.
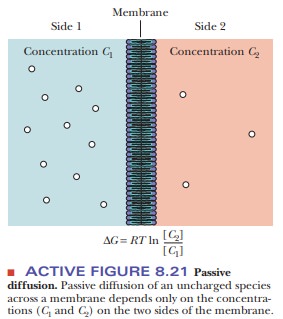
The process of passive transport can be
subdivided into two categories-sim-ple diffusion and facilitated diffusion. In simple diffusion, a molecule moves
directly through the membrane without interacting with another molecule. Small,
uncharged molecules, such as O2, N2, and CO2,
can pass through mem-branes via simple diffusion. The rate of movement through
the membrane is controlled solely by the concentration difference across the
membrane (Figure 8.21). Larger molecules (especially polar ones) and ions
cannot pass through a membrane by simple diffusion. The process of moving a
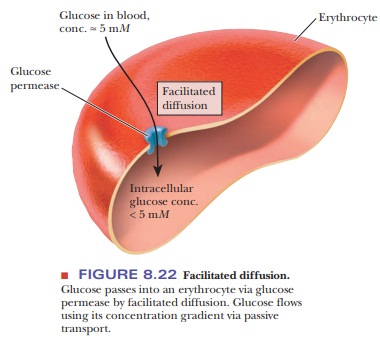
molecule passively through a membrane using a carrier protein, to which
molecules bind, is called facilitated
diffusion. A good example is the movement of glucose into erythro-cytes.
The concentration of glucose in the blood is about 5 mM. The glucose concentration in the erythrocyte is less than 5 mM. Glucose passes through a carrier
protein called glucose permease (Figure 8.22). This process is labeled as
facilitated diffusion because no energy is expended and a protein carrier is
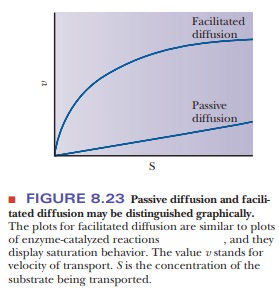
used. In addition, facilitated diffusion is identified by the fact that the
rate of transport, when plotted against the concentration of the molecule being
transported, gives a hyperbolic curve similar to that seen in Michaelis–Menten
enzyme kinetics (Figure 8.23). In a carrier protein, a pore is created by
folding the backbone and side chains. Many of these proteins have several α-helical
portions that span the membrane; in others, a β-barrel forms the pore. In one
example, the helical portion of the protein spans the membrane. The exterior,
which is in contact with the lipid bilayer, is hydrophobic, whereas the
interior, through which ions pass, is hydrophilic. Note that this orientation
is the inverse of that observed in water-soluble globular proteins.
Active transport requires moving substances against a concentration gradi-ent. It is
identified by the presence of a carrier protein and the need for an energy
source to move solutes against a gradient. In primary active transport, the movement of molecules against a
gradient is directly linked to the hydroly-sis of a high-energy molecule, such
as ATP. The situation is so markedly similar to pumping water uphill that one
of the most extensively studied examples of active transport, moving potassium
ions into a cell and simultaneously moving sodium ions out of the cell, is
referred to as the sodium–potassium ion
pump (or Na+/K+ pump).
Under normal circumstances, the concentration
of K+ is higher inside a cell than in extracellular fluids ([K+]inside>
[K+]outside), but the concentration of Na+ is
lower inside the cell than out ([Na+]inside< [Na+]outside).
The energy required to move these ions against their gradients comes from an
exergonic (energy-
releasing) reaction, the hydrolysis of ATP to
ADP and Pi (phosphate
ion). There can be no transport of ions without hydrolysis of ATP. The same
protein appears to serve both as the enzyme that hydrolyzes the ATP (the
ATPase) and as the transport protein; it consists of several subunits. The
reactants and prod-ucts of this hydrolysis reaction-ATP, ADP, and Pi-remain within the cell, and
the phosphate becomes covalently bonded to the transport protein for part of
the process.
The Na+/K+ pump operates
in several steps (Figure 8.24). One subunit of the protein hydrolyzes the ATP
and transfers the phosphate group to an aspartate
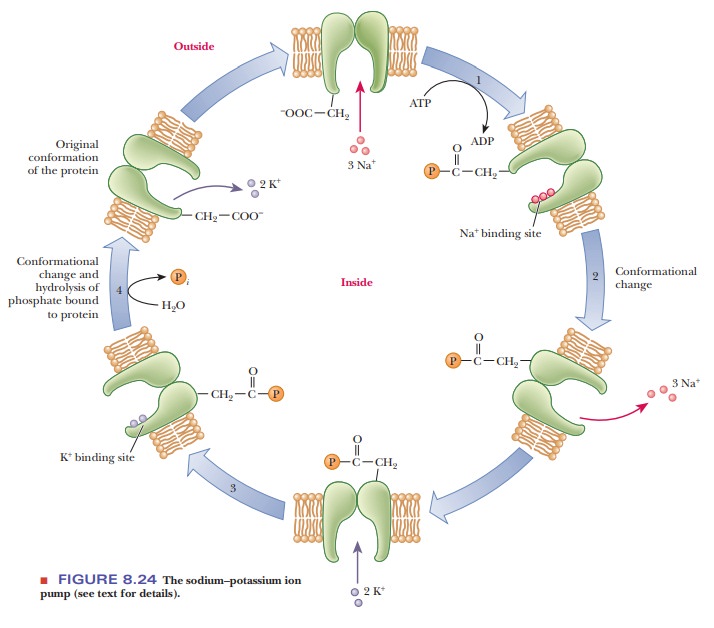
(The
bond formed here is a mixed anhydride;) Simultaneously, binding of three Na+
ions from the interior of the cell takes place. The phosphorylation of one
subunit causes a conformational change in the protein, which opens a channel or
pore through which the three Na+ ions can be released to the
extracellular fluid (step 2). Outside the cell, two K+ ions bind to
the pump enzyme, which is still phosphory-lated (step 3). Another
conformational change occurs when the bond between the enzyme and the phosphate
group is hydrolyzed. This second conformation-al change regenerates the
original form of the enzyme and allows the two K+ ions to enter the
cell (step 4). The pumping process transports three Na+ ions out of
the cell for every two K+ ions transported into the cell (Figure
8.25).
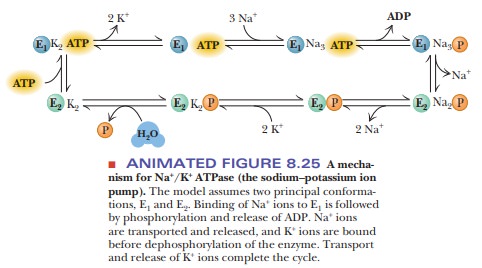
The operation of the pump can be reversed when
there is no K+ and a high concentration of Na+ in the
extracellular medium; in this case, ATP is produced by the phosphorylation of
ADP. The actual operation of the Na+/K+ pump is not
completely understood and probably is even more complicated than we now know.
There is also a calcium ion (Ca2+) pump, which is a sub-ject of
equally active investigation. Unanswered questions about the detailed mechanism
of active transport provide opportunities for future research.
Another type of transport is called secondary active transport. An example is the galactoside permease in bacteria (Figure 8.26). The lactose concentra-tion inside the bacterial cell is higher than the concentration outside, so mov-ing lactose into the cell requires energy. The galactoside permease does not directly hydrolyze ATP, however. Instead, it harnesses the energy by letting hydrogen ions flow through the permease into the cell with their concentration gradient. As long as more energy is available allowing the hydrogen ions to flow (–DG) than is required to concentrate the lactose (+DG), the process is possi-ble.
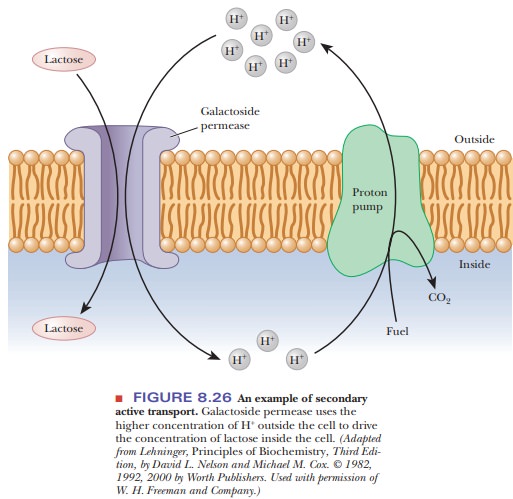
However, to arrive at a situation in which there is a higher concentration of hydrogen
ions on the outside than on the inside, some other primary active transporter
must establish the hydrogen ion gradient. Active transporters that create
hydrogen ion gradients are called proton
pumps.
How do membrane receptors work?
The
first step in producing the effects of some biologically active substances is
binding the substance to a protein receptor site on the exterior of the cell.
The interaction between receptor proteins and the active substances that bind
to them has features in common with enzyme–substrate recognition. There is a
requirement for essential functional groups that have the correct
three-dimensional conformation with respect to each other. The binding site,
whether on a receptor or an enzyme, must provide a good fit for the substrate.
In receptor binding, as in enzyme behavior, inhibition of the action of the
protein by some sort of “poison” or inhibitor is possible. The study of
receptor proteins is less advanced than the study of enzymes because many
receptors are tightly bound integral proteins, and their activity depends on
the membrane environment. Receptors are often large oligomeric proteins (ones
with several subunits), with molecular weights on the order of hundreds of
thousands. Also, quite frequently, the receptor has very few molecules in each
cell, adding to the difficulties of isolating and studying this type of
protein.
An
important type of receptor is that for low-density lipoprotein (LDL), the
principal carrier of cholesterol in the bloodstream. LDL is a particle that
consists of various lipids-in particular, cholesterol and phosphoglycerides-as
well as a protein. The protein portion of the LDL particle binds to the LDL
receptor of a cell. The complex formed between the LDL and the receptor is
pinched off into the cell in a process called endocytosis. The receptor protein is then recycled back to the
surface of the cell (Figure 8.27). The cholesterol portion of the LDL is used
in the cell, but an
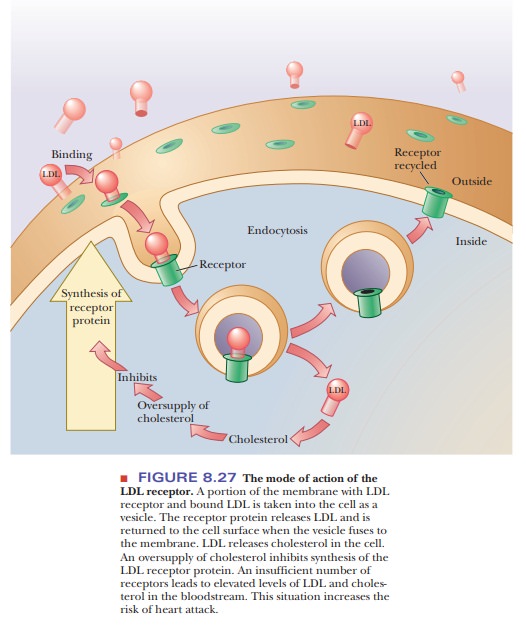
Excess of cholesterol inhibits the synthesis of
LDL receptor. If there are too few receptors for LDL, the level of cholesterol
in the bloodstream increases. Eventually, the excess cholesterol is deposited
in the arteries, blocking them severely. This blocking of arter-ies, called atherosclerosis,
can eventually lead to heart attacks and strokes. In many industrialized
countries, typical blood cholesterol levels are high, and the incidence of
heart attacks and strokes is correspondingly high.
Summary
Molecules are transported across cell membranes in several ways,
and proteins play a role in most of them.
In simple diffusion, small, uncharged molecules
cross the membrane without a carrier protein. In facilitated diffusion,
substances bind to a carrier protein. Neither process requires energy, and the
two together are called passive transport.
In active transport, energy is required, either directly or
indirectly. Large membrane proteins play a key role in the process.
Proteins
serve as receptors for substances that bind to cell surfaces.
Related Topics