Chapter: Biochemistry: Lipids and Proteins Are Associated in Biological Membranes
What are triacylglycerols?
What are triacylglycerols?
Glycerol is a simple compound that contains three hydroxyl groups
(Figure8.2). When all three of the alcohol groups form ester linkages with
fatty acids, the resulting compound is a triacylglycerol;
an older name for this type of compound is triglyceride.
Note that the three ester groups are the polar part of the molecule, whereas
the tails of the fatty acids are nonpolar. It is usual for three different
fatty acids to be esterified to the alcohol groups of the same glycerol
molecule. Triacylglycerols do not occur as components of membranes (as do other
types of lipids), but they accumulate in adipose tissue (primarily fat cells)
and provide a means of storing fatty acids, particularly in animals. They serve
as concentrated stores of metabolic energy. Complete oxidation of fats yields
about 9 kcal g–1, in contrast with 4 kcal g–1 for
carbohydrates and proteins.
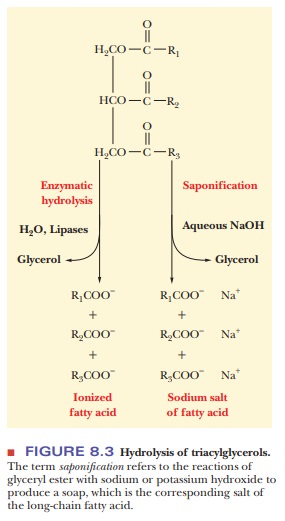
When an organism uses fatty acids, the ester
linkages of triacylglycerols are hydrolyzed by enzymes called lipases. The same hydrolysis reaction
can take place outside organisms, with acids or bases as catalysts. When a base
such as sodium hydroxide or potassium hydroxide is used, the products of the
reac-tion, which is called saponification
(Figure 8.3), are glycerol and the sodium or potassium salts of the fatty
acids. These salts are soaps. When soaps are used with hard water, the calcium
and magnesium ions in the water react with the fatty acids to form a precipitate-the
characteristic scum left on the insides of sinks and bathtubs. The other
product of saponification, glycerol, is used in creams and lotions as well as
in the manufacture of nitroglycerin.
Related Topics