Chapter: 11th Chemistry : UNIT 10 : Chemical bonding
Salient features of Molecular orbital theory
The salient features of this theory are as follows.
1. When atoms combines to form molecules, their individual atomic orbitals lose their identity and forms new orbitals called molecular orbitals.
2. The shapes of molecular orbitals depend upon the shapes of combining atomic orbitals.
3. The number of molecular orbitals formed is the same as the number of combining atomic orbitals. Half the number of molecular orbitals formed will have lower energy than the corresponding atomic orbital, while the remaining molecular orbitals will have higher energy. The molecular orbital with lower energy is called bonding molecular orbital and the one with higher energy is called anti-bonding molecular orbital. The bonding molecular orbitals are represented as σ (Sigma), π (pi), δ (delta) and the corresponding antibonding orbitals are denoted as σ*, π* and δ*.
4. The electrons in a molecule are accommodated in the newly formed molecular orbitals. The filling of electrons in these orbitals follows Aufbau's principle, Pauli's exclusion principle and Hund's rule as in the case of filling of electrons in atomic orbitals.
5. Bond order gives the number of covalent bonds between the two combining atoms. The bond order of a molecule can be calculated using the following equation
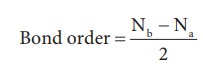
Where,
Nb = Total number of electrons present in the bonding molecular orbitals
Na = Total number of electrons present in the antibonding molecular orbitals and
A bond order of zero value indicates that the molecule doesn't exist.
Related Topics