Chemical bonding - Polarity of Bonds | 11th Chemistry : UNIT 10 : Chemical bonding
Chapter: 11th Chemistry : UNIT 10 : Chemical bonding
Polarity of Bonds
Polarity of Bonds
Partial ionic character in covalent bond:
When a covalent bond is formed between two identical atoms (as in the case of H2, O2, Cl2 etc...) both atoms have equal tendency to attract the shared pair of electrons and hence the shared pair of electrons lies exactly in the middle of the nuclei of two atoms. However, in the case of covalent bond formed between atoms having different electronegativities, the atom with higher electronegativity will have greater tendency to attract the shared pair of electrons more towards itself than the other atom. As a result the cloud of shared electron pair gets distorted.
Let us consider the covalent bond between hydrogen and fluorine in hydrogen fluoride. The electronegativities of hydrogen and fluorine on Pauling's scale are 2.1 and 4 respectively. It means that fluorine attracts the shared pair of electrons approximately twice as much as the hydrogen which leads to partial negative charge on fluorine and partial positive charge on hydrogen. Hence, the H-F bond is said to be polar covalent bond.
![]()
![]() Here, a very small, equal and opposite charges are separated by a small distance (91 pm) and is referred to as a dipole.
Here, a very small, equal and opposite charges are separated by a small distance (91 pm) and is referred to as a dipole.
Dipole moment:
The polarity of a covalent bond can be measured in terms of dipole moment which is defined as
μ = q × 2d
Where μ is the dipole moment, q is the charge and 2d is the distance between the two charges. The dipole moment is a vector and the direction of the dipole moment vector points from the negative charge to positive charge.
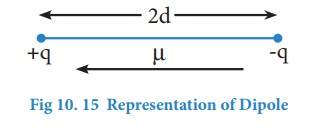
The unit for dipole moment is columb meter (C m). It is usually expressed in Debye unit (D). The conversion factor is 1 Debye = 3.336 x 10-30 C m
Diatomic molecules such as H2, O2, F2 etc... have zero dipole moment and are called non polar molecules and molecules such as HF, HCl, CO, NO etc... have non zero dipole moments and are called polar molecules.
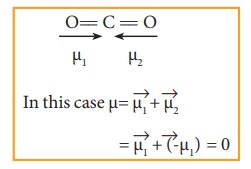
Molecules having polar bonds will not necessarily have a dipole moment. For example, the linear form of carbon dioxide has zero dipole moment, even though it has two polar bonds. In CO2, the dipole moments of two polar bonds (CO) are equal in magnitude but have opposite direction. Hence, the net dipole moment of the CO2 is, μ = μ1 + μ2 = μ1 + (-μ1) = 0
Incase of water net dipole moment is the vector sum of μ1+ μ2 as shown.
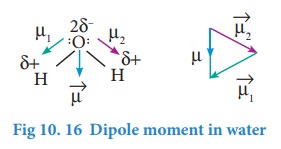
Dipole moment in water is found to be 1.85D
Table 10. 5 Dipole moments of common molecules
The extent of ionic character in a covalent bond can be related to the electro negativity difference to the bonded atoms. In a typical polar molecule, Aδ--Bδ+, the electronegativity difference (χA- χB) can be used to predict the percentage of ionic character as follows.
![]()
![]() If the electronegativity difference (χA- χB ), is
If the electronegativity difference (χA- χB ), is
equal to 1.7, then the bond A-B has 50% ionic character
if it is greater than 1.7, then the bond A-B has more than 50% ionic character, and if it is lesser than 1.7, then the bond A-B has less than 50% ionic character.
Partial covalent character in ionic bonds:
Like the partial ionic character in covalent compounds, ionic compounds show partial covalent character. For example, the ionic compound, lithium chloride shows covalent character and is soluble in organic solvents such as ethanol.
The partial covalent character in ionic compounds can be explained on the basis of a phenomenon called polarisation. We know that in an ionic compound, there is an electrostatic attractive force between the cation and anion. The positively charged cation attracts the valence electrons of anion while repelling the nucleus. This causes a distortion in the electron cloud of the anion and its electron density drifts towards the cation, which results in some sharing of the valence electrons between these ions. Thus, a partial covalent character is developed between them. This phenomenon is called polarisation.
The ability of a cation to polarise an anion is called its polarising ability and the tendency of an anion to get polarised is called its polarisability. The extent of polarisation in an ionic compound is given by the Fajans rules
Fajans Rules
(i) To show greater covalent character, both the cation and anion should have high charge on them. Higher the positive charge on the cation, greater will be the attraction on the electron cloud of the anion. Similarly higher the magnitude of negative charge on the anion, greater is its polarisability. Hence, the increase in charge on cation or in anion increases the covalent character
Let us consider three ionic compounds aluminum chloride, magnesium chloride and sodium chloride. Since the charge of the cation increase in the order Na+ < Mg2+ < Al3+, the covalent character also follows the same order NaCl < MgCl2 < AlCl3.
(ii) The smaller cation and larger anion show greater covalent character due to the greater extent of polarisation.
Lithium chloride is more covalent than sodium chloride. The size of Li+ is smaller than Na+ and hence the polarising power of Li+ is more. Lithium iodide is more covalent than lithium chloride as the size of I- is larger than the Cl-. Hence I- will be more polarised than Cl- by the cation, Li+ .
(iii) Cations having ns2 np6 nd10 configuration show greater polarising power than the cations with ns2 np6 configuration. Hence, they show greater covalent character.
CuCl is more covalent than NaCl. Compared to Na+ (1.13 Å) . Cu+ (0.6 Å) is small and have 3s2 3p6 3d10 configuration.
Electronic configuration of Cu+ [Ar] 3s2, 3p6, 3d10
![]()
![]() Electronic Configuration of Na+ [He] 2s2, 2p6
Electronic Configuration of Na+ [He] 2s2, 2p6
Related Topics