Chemistry - Enthalpy (H) | 11th Chemistry : UNIT 7 : Thermodynamics
Chapter: 11th Chemistry : UNIT 7 : Thermodynamics
Enthalpy (H)
Enthalpy (H)
The enthalpy (H), is a thermodynamic property of a system, is defined as the sum of the internal energy (U) of a system and the product of pressure and volume of the system. That is,
H = U + PV ŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆō(7.8)
It reflects the capacity to do mechanical work and the capacity to release heat by the system. When a process occurs at constant pressure, the heat involved (either released or absorbed) is equal to the change in enthalpy.
Enthalpy is a state function which depends entirely on the state functions T, P and U. Enthalpy is usually expressed as the change in enthalpy (ΔH) for a process between initial and final states at constant pressure.
ŌłåH = ŌłåU + PŌłåV ŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆō(7.9)
The change in enthalpy (ŌłåH) is equal to the heat supplied at the constant pressure to a system (as long as the system does no additional work).
ŌłåH=qp
In an endothermic reaction heat is absorbed by the system from the surroundings that is q>0 (positive). Therefore, ΔH is also positive. In an exothermic reaction heat is evolved by the system to the surroundings that is, q<0 (negative). If q is negative, then ΔH will also be negative.
Relation between enthalpy `H' and internal energy `U'
When the system at constant pressure undergoes changes from an initial state with H1, U 1 and V 1 to a final state with H2, U2 and V2the change in enthalpy ΔH, can be calculated as follows:
H=U + PV
In the initial state
H1=U1 + PV1ŌłÆŌłÆŌłÆŌłÆŌłÆ(7.10)
In the final state
H2 = U2 + PV2ŌłÆŌłÆŌłÆŌłÆ(7.11)
change in enthalpy is (7.11) - (7.10)
(H2ŌłÆH1) = (U2ŌłÆU1) + P(V2ŌłÆV1)
ŌłåH=ŌłåU + PŌłåV --------- (7.12)
As per first law of thermodynamics,
ŌłåU = q+w
Equation 7.12 becomes
ŌłåH = q + w + PŌłåV
w= -PŌłåV
ŌłåH = qp - PŌłåV + PŌłåV
ŌłåH = qpŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆō (7.13)
qpŌłÆ is the heat absorbed at constant pressure and is considered as heat content.
Consider a closed system of gases which are chemically reacting to form gaseous products at constant temperature and pressure with Vi and Vf as the total volumes of the reactant and product gases respectively, and ni and nf as the number of moles of gaseous reactants and products, then,
For reactants (initial state) :
PVi = ni RT ---------(7.14)
For products (final state) :
PVf = nf RT --------- (7.15)
(7.15) - (7.14)
(Vf -Vi) = (nf - ni) RT
PŌłåV= Dn(g) RT ŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆō(7.16)
Substituting in 7.16 in 7.12
ŌłåH = ŌłåU +Dn(g) RTŌĆōŌĆōŌĆōŌĆōŌĆōŌĆōŌĆō(7.17)
Enthalpy Changes for Different Types of Reactions and Phase Transitions:
The heat or enthalpy changes accompanying chemical reactions is expressed in different ways depending on the nature of the reaction. These are discussed below.
Standard heat of formation
The standard heat of formation of a compound is defined as ŌĆ£the change in enthalpy that takes place when one mole of a compound is formed from its elements, present in their standard states (298 K and 1 bar pressure)". By convention the standard heat of formation of all elements is assigned a value of zero.
Fe(s)+ S(s) ŌåÆ FeS(s)
╬öHf0 = ŌĆō 100.42 kJ molŌłÆ1
2C(s)+H2(g)ŌåÆ C2H2(g)
╬öHf0= + 222.33 kJ molŌłÆ1
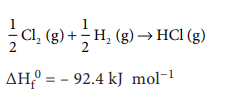
╬öHf0 = ŌĆō 92.4 kJ molŌłÆ1
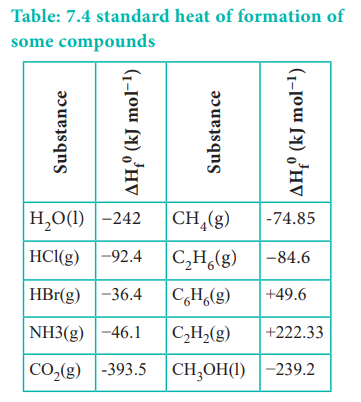
The standard heats of formation of some compounds are given in Table 7.4.
Related Topics