Chapter: Modern Analytical Chemistry: Titrimetric Methods of Analysis
Titrations Based on Acid–Base Reactions: Qualitative Applications
Qualitative Applications
We have already come across one example of the qualitative application of acid–base titrimetry in assigning the forms of alkalinity in waters (see Example 9.5).


This approach is easily extended to other systems.
For example, the composition of solutions containing one or two of the following species
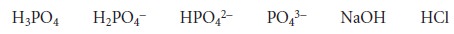
can
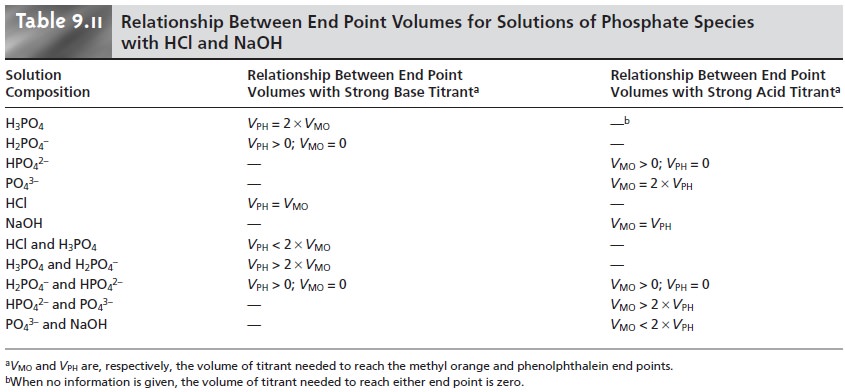
be determined by titrating with either a strong acid or a strong base to the methyl orange and phenolphthalein end points. As outlined in Table 9.11,
each species or mixture
of species has a unique
relationship between the volumes of titrant needed to reach
these two end points.
Related Topics