Chapter: Modern Analytical Chemistry: Titrimetric Methods of Analysis
Evaluation of Redox Titrimetry
Evaluation of Redox Titrimetry
The
scale of operations, accuracy, precision, sensitivity, time, and cost of
methods involving redox
titrations are similar
to those described earlier for acid–base
and complexometric titrimetric methods. As with acid–base titrations, redox titrations can be extended
to the analysis of mix- tures if there is a significant difference in the ease with which the analytes can be
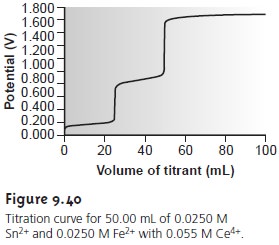
oxidized or reduced. Figure 9.40 shows
an example of the titration curve for a mixture
of Fe2+
and Sn2+, using
Ce4+ as
the titrant. The titration of a
mixture of analytes whose standard-state potentials or formal potentials differ
by at least 200 mV will result in a separate equivalence point for each
analyte.
Related Topics