Chapter: Modern Analytical Chemistry: Titrimetric Methods of Analysis
Evaluation of Complexation Titrimetry
Evaluation of Complexation Titrimetry
The scale of operations, accuracy,
precision, sensitivity, time, and cost of methods involving complexation titrations are similar to those described earlier for acid–base titrimetric methods. Compared
with acid–base titrations, however, complexation titrations are more selective. Despite the ability
of EDTA to form strong complexes
with virtually all
metal ions, carefully controlling the pH at which
the titration is carried out makes it possible to
analyze samples containing two or more analytes
(see Example 9.10). The reason
that pH can
be used to provide selectivity is easily
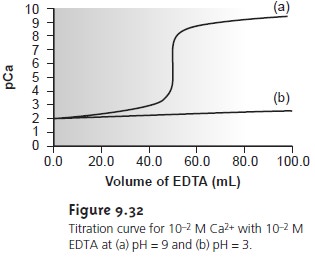
appreciated by examining Figure 9.32. A titration of Ca2+ at a pH of 9 gives
a dis- tinct break in the titration curve because the conditional formation
constant (K´f = 2.6 x 109) is large
enough to ensure
that the reaction
of Ca2+
and EDTA goes to
completion. At a pH of 3, however,
the conditional formation
constant (K´f =
1.23) is so small
that very little
Ca2+ reacts with the EDTA.



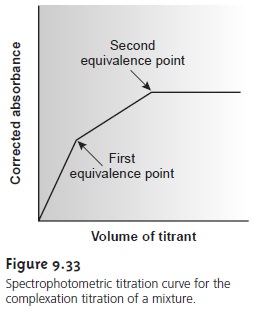
Spectrophotometric titrations are particularly useful
for the analysis
of mixtures if a suitable difference in absorbance exists between the analytes and products, or titrant. For example, the analysis of a two-component mixture can be accomplished
if there is a difference between the absorbance of the two metal–ligand complexes (Figure 9.33).
Related Topics