Chapter: Modern Analytical Chemistry: Titrimetric Methods of Analysis
Overview of Titrimetry
Overview of Titrimetry
Titrimetric methods are classified into four groups based on the type of reaction
in- volved. These groups are acid–base
titrations, in which an acidic or basic titrant re-
acts with an analyte that is a base or an acid;
complexometric titrations involving a metal–ligand complexation reaction;
redox titrations, where the titrant
is an oxidiz- ing or reducing
agent; and precipitation titrations, in which the analyte
and titrant react to form a precipitate. Despite
the difference in chemistry, all titrations share several common features, providing the focus for this section.
Equivalence Points and End Points
For a titration to be accurate
we must add a stoichiometrically equivalent amount of titrant
to a solution containing the
analyte. We call
this stoichiometric mixture the equivalence point.
Unlike precipitation gravimetry, where the precipitant is added in excess,
determining the exact
volume of titrant
needed to reach
the equiv- alence point is essential. The product of the equivalence point volume, Veq, and the
titrant’s concentration, CT, gives
the moles of titrant reacting
with the analyte.
Moles titrant = Veq x CT
Knowing the stoichiometry of the titration reaction(s), we can
calculate the moles of analyte.
Unfortunately, in most titrations we
usually have no obvious indication that the
equivalence point has
been reached. Instead, we stop adding
titrant when we reach an end point of
our choosing. Often
this end point
is indicated by a change
in the color of a substance added to the solution containing the analyte. Such sub-
stances are known as indicators. The difference between
the end point volume and the
equivalence point volume
is a determinate method error,
often called the titra- tion error. If the end point
and equivalence point
volumes coincide closely,
then the titration error
is insignificant and can be safely ignored.
Clearly, selecting an ap-
propriate end point is critical
if a titrimetric method is to give accurate results.
Volume as a Signal*
Almost
any chemical
reaction can serve as a titrimetric method provided that three conditions are met. The first condition
is that all reactions involving
the titrant and
analyte must be of known
stoichiometry. If this
is not the
case, then the moles
of titrant used
in reaching the
end point cannot
tell us how
much ana- lyte is in our
sample. Second, the
titration reaction must
occur rapidly. If we add titrant at a rate
that is faster
than the reaction’s rate, then the
end point will
ex- ceed the equivalence point by a significant amount.
Finally, a suitable
method must be available for determining the end point
with an acceptable level of accu- racy. These are significant limitations and, for this reason,
several titration strate- gies are commonly used.
A
simple example of a titration is an analysis
for Ag+
using thiocyanate, SCN–, as a titrant.

This reaction occurs
quickly and is of known
stoichiometry. A titrant
of SCN– is easily prepared using KSCN.
To indicate the titration’s end point we add a small
amount of Fe3+ to the
solution containing the
analyte. The formation of the red- colored Fe(SCN)2+ complex signals
the end point. This is an example
of a direct titration since the titrant reacts
with the analyte.
If
the titration reaction
is too slow, a suitable
indicator is not available, or there
is no useful direct titration reaction, then an indirect analysis
may be possible. Sup- pose you wish to determine the concentration of formaldehyde, H2CO, in an aque- ous solution. The oxidation of H2CO by I3–
H2CO(aq) + 3OH–(aq)+I3–(aq) < == == > HCO3–(aq)+ 3I–(aq)+ 2H2O(l)
is a useful
reaction, except that
it is too slow for
a direct titration. If we add
a known amount of I3–, such that it is in excess, we can allow
the reaction to go to comple-
tion. The I3– remaining can
then be titrated with thiosulfate, S2O32–.
I3–(aq)+ 2S2O32–(aq) < == == > S4O62–(aq)+ 3I–(aq)
This type of titration is called a back titration.
Calcium ion plays
an important role in many aqueous environmental systems. A useful direct analysis takes advantage of its reaction
with the ligand ethylenedi-
aminetetraacetic acid (EDTA),
which we will represent as Y4–.
Ca2+(aq)+
Y4–(aq) < == > CaY2–(aq)
Unfortunately, it often
happens that there
is no suitable indicator for
this direct titration. Reacting Ca2+ with an excess of the Mg2+–EDTA complex
Ca2+(aq)
+ MgY2–(aq) < == == > CaY2–(aq)+ Mg2+(aq)
releases an equivalent amount of Mg2+.
Titrating the released Mg2+ with EDTA
Mg2+(aq)+ Y4–(aq) < == == > MgY2–(aq)
gives a suitable
end point. The amount of Mg2+ titrated provides an indirect
mea- sure of the amount of Ca2+ in
the original sample.
Since the analyte
displaces a species that
is then titrated, we call this
a displacement titration.
When a suitable
reaction involving the analyte does not exist
it may be possible to generate
a species that is easily
titrated. For example,
the sulfur content
of coal can be
determined by using
a combustion reaction
to convert sulfur
to sulfur dioxide.
S(s)+ O2(g) → SO2(g)
Passing the SO2 through an aqueous
solution of hydrogen peroxide, H2O2,
SO2(g)+ H2O2(aq)
→ H2SO4(aq)
produces sulfuric acid, which we can titrate with NaOH,
H2SO4(aq) + 2OH–(aq)
< == == > SO42–(aq)+ 2H2O(l)
providing an indirect determination of sulfur.
Titration Curves
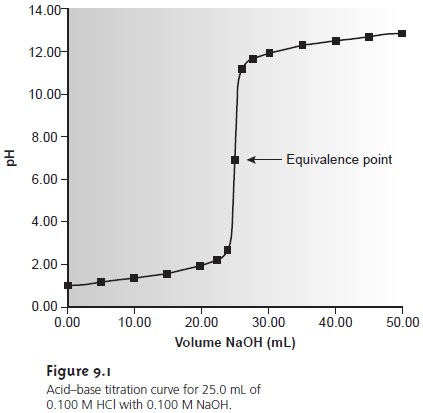
To find the end point we monitor some property of the titration reaction that has a well-defined value at the equivalence point. For example, the equivalence point for a titration of HCl with NaOH occurs at a pH of 7.0. We can find the end point, therefore, by monitoring the pH with a pH electrode or by adding an indicator that changes color at a pH of 7.0.
Suppose that the
only available indicator changes color at a pH of 6.8.
Is this end point
close enough to the equivalence point that the titration error
may be safely ignored?
To answer this question we need to know how the pH changes dur- ing the titration.
A titration
curve provides us with a visual picture
of how a property, such
as pH, changes as we add titrant (Figure
9.1). We can measure this titration curve ex-
perimentally by suspending a pH electrode
in the solution containing the analyte,
monitoring the pH as titrant
is added. As we will see later, we can also calculate
the expected titration curve
by considering the reactions responsible for the change
in pH. However we arrive at the titration curve, we may use it to evaluate
an indica- tor’s likely
titration error. For
example, the titration curve in Figure
9.1 shows us that
an end point
pH of 6.8 produces a small titration error. Stopping the
titration at an end point pH of 11.6,
on the other hand, gives
an unacceptably large
titration error.
The
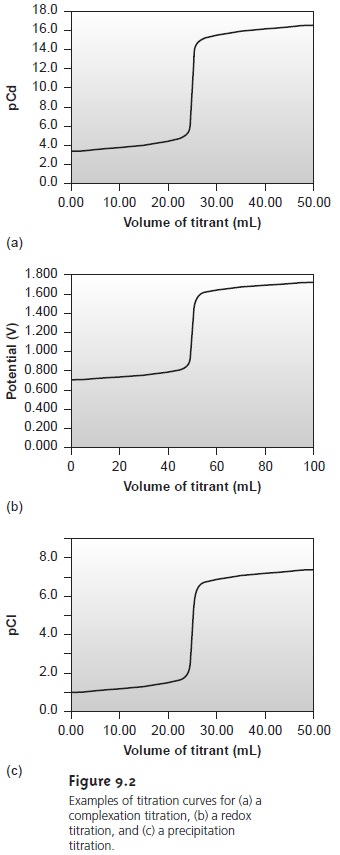
titration curve in Figure 9.1 is not unique to an acid–base titration.
Any titration curve
that follows the change in concentration of a species
in the titration reaction
(plotted logarithmically) as a function
of the volume of titrant has the same general sigmoidal
shape. Several additional examples are shown in Figure 9.2.
Concentration is not the
only property that
may be used
to construct a titration
curve. Other parameters, such as temperature or the absorbance of light, may be
used if they show a significant change
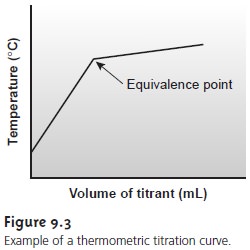
in value at the equivalence point. Many titra- tion reactions, for example,
are exothermic. As the titrant
and analyte react,
the temperature of the system steadily
increases. Once the titration is complete, further additions of titrant do not produce
as exothermic a response, and the change
in temperature levels off.
A typical titration curve of temperature versus volume of titrant is shown in Figure 9.3. The titration
curve contains two linear segments,
the intersection of which
marks the equivalence point.
The Buret
The
only essential piece
of equipment for an acid–base titration is a means for deliv-
ering the titrant to the solution containing the analyte. The most common method
for delivering the titrant is a buret (Figure 9.4). A buret
is a long, narrow tube with
graduated markings, and
a stopcock for
dispensing the titrant. Using a buret
with a small internal
diameter provides a better defined
meniscus, making it easier to read
the buret’s volume precisely. Burets
are available in a variety
of sizes and
tolerances
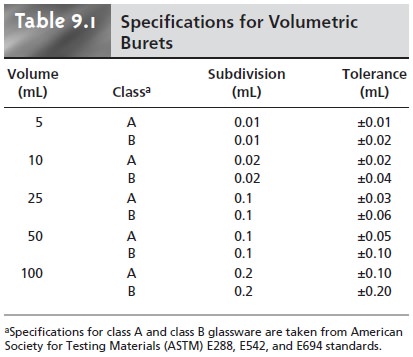
(Table 9.1), with
the choice of buret determined by the demands
of the analysis. The accuracy obtainable with a buret can be improved by calibrating it over several intermediate ranges of volumes
using the same method described for calibrating pipets.
In this manner, the volume of titrant
delivered can be corrected
for any variations in the buret’s internal
diameter.
Titrations may be automated using a pump to deliver
the titrant at a constant flow rate, and a solenoid valve
to control the flow (Figure
9.5). The volume
of titrant delivered is determined by multiplying the flow rate by the elapsed time.
Au- tomated titrations offer the additional advantage of using a microcomputer for data storage and analysis.

Related Topics