Chapter: Modern Analytical Chemistry: Chromatographic and Electrophoretic Methods
Optimizing Chromatographic Separations
Optimizing
Chromatographic Separations
Now that we have defined
capacity factor, selectivity, and column efficiency we con- sider their
relationship to chromatographic resolution. Since we are only interested
in the resolution between solutes
eluting with similar
retention times, it is safe
to as- sume that
the peak widths
for the two
solutes are approximately the same. Equation 12.1, therefore, is written
as
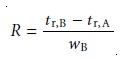 12.19
12.19
Solving equation 12.17
for wB and substituting into equation 12.19
gives
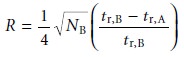 12.20
12.20
The retention times for solutes
A and B are replaced
with their respective capacity factors by rearranging equation 12.10
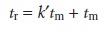
and substituting into equation 12.20.
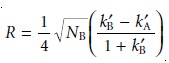
Finally, solute A’s
capacity factor is eliminated using
equation 12.11. After
rearrang- ing, the equation
for the resolution between the chromatographic peaks for solutes A and B is
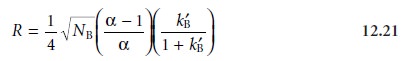
Besides resolution, another
important factor in chromatography is the amount of time required to elute a pair of solutes. The time needed
to elute solute
B is
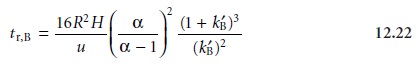
Equations 12.21 and 12.22 contain
terms corresponding to column efficiency, column selectivity, and capacity
factor. These terms
can be varied, more or less in- dependently, to obtain the desired resolution and analysis time for a pair of solutes.
The first term, which is a function
of the number of theoretical plates or the height
of a
theoretical plate, accounts
for the effect of column efficiency. The second term is
a function of α and accounts for the influence of column selectivity. Finally, the third term in both equations is a function
of kB’, and accounts for the effect
of solute B’s capacity
factor. Manipulating these
parameters to improve
resolution is the sub-
ject of the remainder of this section.
Related Topics