Chapter: Modern Analytical Chemistry: Chromatographic and Electrophoretic Methods
Gas Chromatography: Qualitative Applications
Qualitative Applications
Gas chromatography also can be used for qualitative purposes. When using an FT–IR
or a mass spectrometer as the detector, the available spectral information often can be used to identify individual solutes.
With conventional nonspectroscopic detectors, other methods
must be used to identify the solutes. One approach is to spike
the sample by adding an aliquot of a
suspected analyte and looking for an increase
in peak height. Retention times also
can be compared with values
measured for standards, provided that the operating
conditions are identical. Because of the difficulty of exactly matching such condi- tions, tables of retention times are of limited utility.
Kovat’s retention index provides one solution to the matching of retention times. Under
isothermal conditions, the adjusted retention times of normal
alkanes increase logarithmically. Kovat
defined the retention index, I, for a normal alkane
as 100 times the number of carbon atoms; thus, the retention index is 400 for butane and 500 for pentane. To determine the
retention index for
another compound, its adjusted retention time is measured relative
to that for the normal
alkanes eluting just before
and after. For example, a compound eluting between butane and pen- tane has a retention index between 400 and 500. The exact
value for the com-
pound’s retention index,
Icpd, is given as
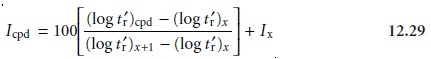
where x is the
normal alkane eluting
before the compound, and x + 1 is the normal alkane eluting just after
the compound.

Related Topics