Chapter: Modern Analytical Chemistry: Chromatographic and Electrophoretic Methods
Column Efficiency - Theory of Column Chromatography
Column Efficiency
At the beginning of a chromatographic separation the solute
occupies a narrow band of finite width. As the solute passes through the column, the width of its band continually increases in a process
called band broadening. Column
efficiency pro- vides a quantitative measure of the extent of band broadening.
In their original
theoretical model of chromatography, Martin and Synge2 treated the
chromatographic column as though it consists of discrete sections at which partitioning of the solute between the stationary and mobile phases occurs.
They
called each section a theoretical plate
and defined column efficiency in terms of the number
of theoretical plates,
N, or the height
of a theoretical plate, H; where
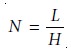 12.12
12.12
A columnŌĆÖs efficiency improves with an increase in the number of theoretical plates or a decrease in the height
of a theoretical plate.
Assuming a Gaussian
profile, the extent
of band broadening is measured by the
variance or standard deviation of a chromatographic peak. The height
of a theoreti- cal plate is defined as the variance
per unit length
of the column
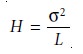 12.13
12.13
where the variance,
Žā2, has units of distance squared.
Because retention time and
peak width are usually measured in seconds or minutes, it is more
convenient to ex- press the standard deviation in units of time, Žä, by dividing
by the mobile
phaseŌĆÖs average linear velocity.
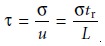 12.14
12.14
When a chromatographic peak has a Gaussian shape,
its width at the baseline, w, is four times its standard deviation, Žä.
w = 4Žä ŌĆ”ŌĆ”ŌĆ”ŌĆ”ŌĆ”ŌĆ”ŌĆ”12.15
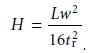 12.16
12.16
The number of theoretical plates in a chromatographic column is obtained
by com- bining equations 12.12 and 12.16.
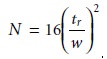 12.17
12.17
Alternatively, the number of theoretical plates can be
approximated as
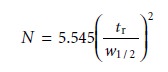
where w1/2
is the width of the chromatographic peak at half its height.

It is important to remember that a theoretical plate is an artificial construct and that no such plates exist
in a chromatographic column. In fact, the number of theo-
retical plates depends on both the properties of the column and the solute. As a re- sult,
the number of theoretical plates
for a column is not fixed and may vary from
solute to solute.
Related Topics