Structure, Preparation - Inter halogen compounds | 12th Chemistry : UNIT 3 : p-Block Elements-II
Chapter: 12th Chemistry : UNIT 3 : p-Block Elements-II
Inter halogen compounds
Inter
halogen compounds:
Each halogen combines
with other halogens to form a series of compounds called inter halogen
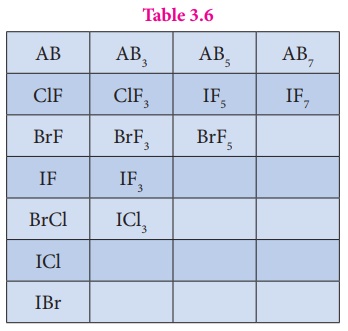
compounds. In the given table of inter halogen compounds a given compound A is
less electronegative than B.
Table 3.6
Properties of inter halogen compounds:
i.
The central atom will be the larger one
ii.
It can be formed only between two halogen and not more than two
halogens.
iii.
Fluorine can’t act as a central metal atom being the smallest one
iv.
Due to high electronegativity with small size fluorine helps the
central atom to attain high coordination number
v.
They can undergo the auto ionization.
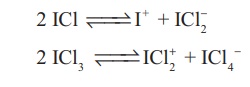
vi. They are strong oxidizing agents
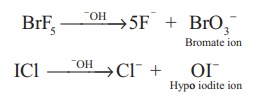
Reaction with alkali:
When heated with the
alkalis, larger halogen form oxyhalogens and the smaller forms halide.
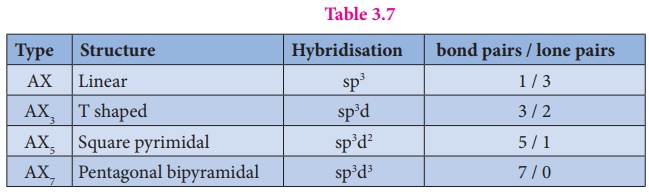
Structure of inter halogen compounds:
The structures of
different type of interhalogen compunds can be easily explained using VSEPR
theory. The details are given below.
Table 3.7
Related Topics