Occurrence, Preparation, Properties, Structure, Uses - Group 18 (Inert gases) elements | 12th Chemistry : UNIT 3 : p-Block Elements-II
Chapter: 12th Chemistry : UNIT 3 : p-Block Elements-II
Group 18 (Inert gases) elements
Group
18 (Inert gases) elements:
Occurrence:
All the noble gases
occur in the atmosphere.
Physical properties:
As we move along the
noble gas elements, their atomic radius and boiling point increases from helium
to radon. The first ionization energy decreases from helium to radon. Noble
gases have the largest ionisation energy compared to any other elements in a
given row as they have completely filled orbital in their outer most shell.
They are extremely stable and have a small tendency to gain or lose electrons.
The common physical properties of the group 18 elements are listed in the
Table.
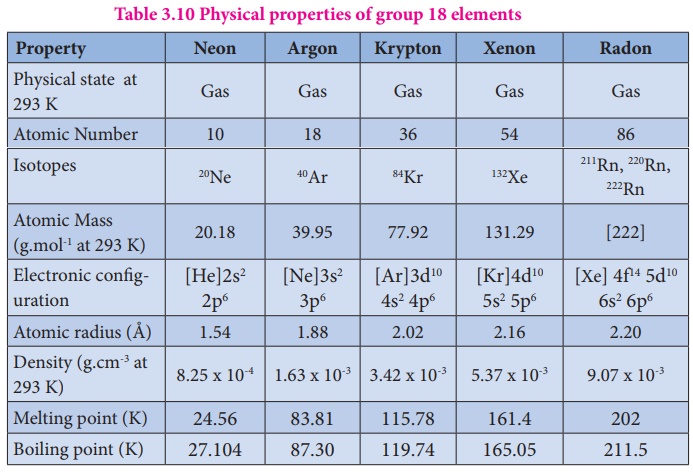
Table 3.10 Physical properties of group 18 elements
Properties of inert gases:
Physical properties:
Noble gases are
monoatomic, odourless, colourless, tasteless, and non-inflammable. They are
highly unreactive. They are non-metallic in nature.
Chemical Properties:
Only the xenon and
krypton show some chemical reactivity. Xenon fluorides are prepare by direct
reaction of xenon and fluorine under different conditions as shown below.
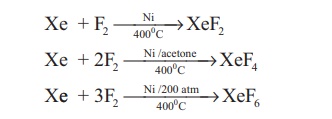
When XeF6 is
heated at 50 °C in a sealed quartz vessel it forms XeOF4.
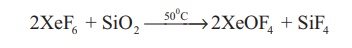
2XeF6 + SiO2
→50 0 C →2XeOF4 + SiF4
When the reaction is
continued the following reaction takes place.
2XeOF4 + SiO2
→ 2XeO2F2
+ SiF4
2XeO2F2
+ SiO2 → 2XeO3 + SiF4
On hydrolysis with water
vapour XeF6 gives XeO3
XeF6 + 3H2O
→ XeO3 + 6HF
When XeF6
reacts with 2.5 M NaOH, sodium per xenate is obtained.
2XeF6 +
16NaOH → Na4XeO6
+ Xe + O2 + 12NaF + 8H2O
Sodium per xenate is
very much known for its strong oxidizing property. For example, it oxidises
manganese (II) ion into permanganate ion even in the absence of the catalyst.
5XeO64− + 2Mn2+ +
14H+ → 2MnO4− + 5XeO3 + 7H2O
Xenon reacts with PtF6
and gave an orange yellow solid [XePtF6] and this is insoluble in
CCl4.
Xenon difluoride forms
addition compounds XeF2.2SbF5 and XeF2.2TaF5.
Xenon hexa fluorides forms compound with boron and alkali metals. Eg : XeF6.BF3,
XeF6MF, M-alkali metals.
There is some evidence
for existence of xenon dichloride XeCl2.
Krypton form krypton difluoride
when an electric discharge is passes through Kr and fluorine at 183° C or when
gases are irradiated with SbF5 it forms KrF2.2SbF3.
Table 3.11 Structures of compounds of Xenon:
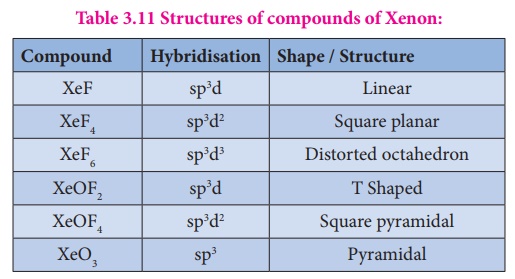
Uses of noble gases:
The inertness of noble
gases is an important feature of their practical uses.
Helium:
·
Helium and oxygen mixture is used by divers in place of air oxygen
mixture. This prevents the painful dangerous condition called bends.
·
Helium is used to provide inert atmosphere in electric arc welding
of metals
·
Helium has lowest boiling point hence used in cryogenics (low
temperature science).
·
It is much less denser than air and hence used for filling air
balloons
Neon:
Neon is used in
advertisement as neon sign and the brilliant red glow is caused by passing
electric current through neon gas under low pressure.
Argon:
Argon prevents the
oxidation of hot filament and prolongs the life in filament bulbs
Krypton:
Krypton is used in
fluorescent bulbs, flash bulbs etc...
Lamps filed with krypton
are used in airports as approaching lights as they can penetrate through dense
fog.
Xenon:
Xenon is used in
fluorescent bulbs, flash bulbs and lasers.
Xenon emits an intense
light in discharge tubes instantly. Due to this it is used in high speed
electronic flash bulbs used by photographers
Radon:
Radon is radioactive and
used as a source of gamma rays
Radon gas is sealed as
small capsules and implanted in the body to destroy malignant i.e. cancer
growth
Related Topics