Solved Example Problems with answers - Evaluate Yourself: Chemistry: Solutions | 11th Chemistry : UNIT 9 : Solutions
Chapter: 11th Chemistry : UNIT 9 : Solutions
Evaluate Yourself: Chemistry: Solutions
Chemistry: Solutions
Evaluate Yourself
1) If 5.6 g of KOH is present in (a) 500 mL and (b) 1 litre of solution, calculate the molarity of each of these solutions.
Solution:
mass of KOH = 5.6 g
no. of moles = 5.6/56 = 0.1 mol
molarity = number of moles of solute / volume of solution (in L)
i) Volume of the solution = 500 ml = 0.5 L
molarity = number of moles of solute / volume of solution (in L)
Molarity = 0.1/0.5 = 0.2M
ii) Volume of the solution = 1 L
molarity = number of moles of solute / volume of solution (in L)
Molarity = 0.1/1 = 0.1M
2) 2.82 g of glucose is dissolved in 30 g of water. Calculate the mole fraction of glucose and water.
Solution:
mass of glucose = 2.82 g
no. of moles of glucose = 2.82/180=0.016
mass of water = 30 g
=30/18 = 1.67
XH20 = 1.67 / [1.67+0.016] = 1.67 / 1.686 = 0.99
XH20 + Xglucose =1
0.99 + Xglucose =1
Xglucose = 1 - 0.99
Xglucose = 0.01
3) The antiseptic solution of iodopovidone for the use of external application contains 10 % w/v of iodopovidone. Calculate the amount of iodopovidone present in a typical dose of 1.5 mL.
Solution:
10% W/V means that 10g of solute in 100ml solution.
amount of iodopovidone in 1.5 ml = 10g/100ml x 1.5ml = 0.15g
4) A litre of sea water weighing about 1.05 kg contains 5 mg of dissolved oxygen (O2). Express the concentration of dissolved oxygen in ppm.
Solution:
[ mass of dissolved solid / mass of water ] x 106
5x10-3/1.05x103 x 106 = 4.76ppm
5) Describe how would you prepare the following solution from pure solute and solvent
(a) 1 L of aqueous solution of 1.5 M CoCl2.
(b) 500 mL of 6.0 % (V/V) aqueous methanol solution.
Solution:
(a) mass of 1.5 moles of CoCl2 = 1.5 ├Ś 129.9
= 194.85 g
194.85 g anhydrons cobalt chloride is dissolved in water and the solution is make up to one litre in a standard flask.
b. 6% V/Vaqeous solution contains 6g of methanol in 100 ml solution.
Ōł┤ To prepare 500 ml of 6% V/V solution of methanol 30g methanol is taken in a 500 ml standard flask and required quantity of water is added to make up the solution to 500 ml.
6) How much volume of 6 M solution of NaOH is required to prepare 500 mL of 0.250 M NaOH solution.
Solution:
C1V1 = C2V2
6 M (V1) = 0.25 M ├Ś 500 ml
V1 = 0.25x500/6
V1 = 20.83 mL
7) Calculate the proportion of O2 and N2 dissolved in water at 298 K. When air containing 20% O2 and 80% N2 by volume is in equilibrium with it at 1 atm pressure. HenryŌĆÖs law constants for two gases are KH(O2) = 4.6 x 104 atm and KH (N2) = 8.5 x 104 atm.
Solution:

8) Explain why the aquatic species are more comfortable in cold water during winter season rather than warm water during the summer.
9) Calculate the mole fractions of benzene and naphthalene in the vapour phase when an ideal liquid solution is formed by mixing 128 g of naphthalene with 39 g of benzene. It is given that the vapour pressure of pure benzene is 50.71 mmHg and the vapour pressure of pure naphthalene is 32.06 mmHg at 300 K.
Solution:
PmmHgPmmHg
P┬║Pure benzene = 50.71 mm Hg
P┬║naphthalene = 32.06 mm Hg
Number of moles of benzene = 39/78 = 0.5 mol
Number of moles of napthalene = 128/128 = 1 mol
mole fraction of benzene = 0.5/1.5 = 0.33
mole fraction of napthalene = 1 ŌĆō 0.33 = 0.67
Partial vapour pressure of benzene = P┬║benzene ├Ś mole fraction of benzene
= 50.71 ├Ś 0.33
= 16.73 mm Hg
Partial vapour pressure of napthalene = 32.06 ├Ś 0.67
= 21.48 mm Hg
Mole fraction of benzene in vapour phase =
= 16.73/[16.73+21.48] = 16.73/38.21 = 0.44
Mole fraction of napthalene in vapour phase = 1 ŌĆō 0.44 = 0.56
10) Vapour pressure of a pure liquid A is 10.0 torr at 27┬░C . The vapour pressure is lowered to 9.0 torr on dissolving one gram of B in 20 g of A. If the molar mass of A is 200 then calculate the molar mass of B.
Solution:
PA┬║ = 10 torr , Psolution = 9 torr
WA = 20 g
WB = 1 g
MA = 200 g molŌĆō1
MB = ?
ŌłåP/ PA┬║ = WBxMA / MBxWA
[10-9]/10 = [1x200]/MBx200
MB= 200/20 x 10 = 100g mol-1
11) 2.56 g of Sulphur is dissolved in 100g of carbon disulphide. The solution boils at 319. 692 K . What is the molecular formula of Sulphur in solution The boiling point of CS2 is 319. 450K. Given that Kb for CS2 = 2.42 K Kg mol-1
Solution:
W2 = 2.56 g
W1 = 100 g
T = 319.692 K
Kb = 2.42 K Kg molŌĆō1
ŌłåTb = (319.692 ŌĆō 319.450) K
= 0.242 K
M2 = KbxW2x100 / ŌłåTbxW1
= 2.42x2.56x1000 / 0.242x100
M2 = 256g mol-1
Molecular mass of sulphur in solution = 256 g molŌĆō1
atomic mass of one mole of sulphur atom = 32
No. of atoms in a molecule of sulphur =256/32=8
Hence molecular formula of sulphur is S8.
12) 2g of a non electrolyte solute dissolved in 75 g of benzene lowered the freezing point of benzene by 0.20 K. The freezing point depression constant of benzene is 5.12 K Kg mol-1. Find the molar mass of the solute.
Solution:
W2 = 2g
W1 = 75 g
ΔTf = 0.2 K
Kf = 5.12 K Kg molŌĆō1
M2 = ?
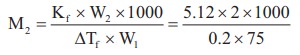
M2= 682.66 mol-1
13) What is the mass of glucose (C6H12O6) in it one litre solution which is isotonic with 6 g L-1 of urea (NH2CONH2) ?
Solution:
Osmotic pressure of urea solution (ŽĆ1)=CRT

14) 0.2 m aqueous solution of KCl freezes at -0.68┬║C calculate vanŌĆÖt Hoff factor. kf for water is 1.86 K kg mol-1.
Solution:
i = observed property / Theoritical property (calculated)
Given ΔTf = 0.680 K
m = 0.2 m
ΔTf (observed) = 0.680 K
ΔTf (calculated) = Kf . m
= 1.86 K Kg molŌĆō1 ├Ś 0.2 mol KgŌĆō1
= 0.372 K
i = (ŌłåTf) observed / (ŌłåTf) calculated = 0.680 K / 0.372 K = 1.82
Related Topics