Chapter: Modern Analytical Chemistry: Spectroscopic Methods of Analysis
Transmittance and Absorbance - Spectroscopy Based on Absorption
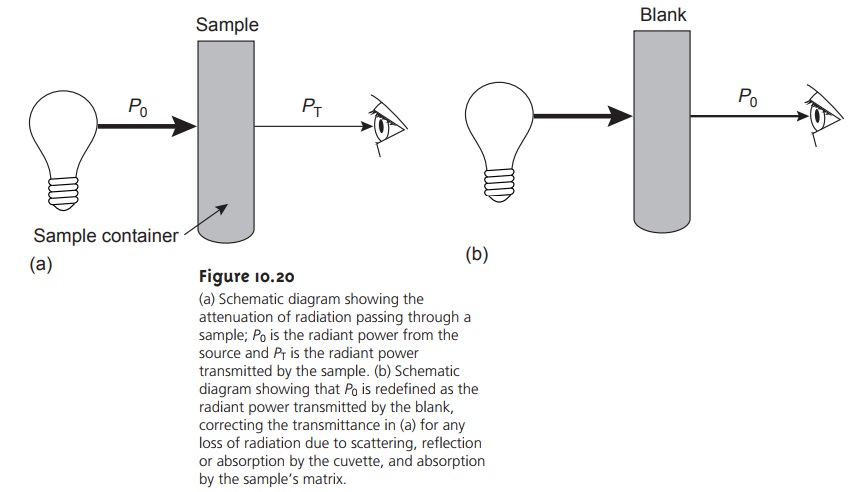
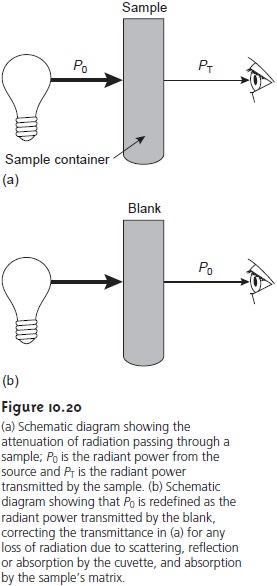
Transmittance and Absorbance
The attenuation of electromagnetic radiation
as it passes through a sample is described quantitatively by two separate, but related terms:
transmittance and absorbance. Transmittance is
defined as the ratio of the electromagnetic radi- ation’s power exiting
the sample, PT, to that incident
on the sample from the source, P0, (Figure
10.20a).
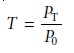 10.1
10.1
Multiplying the transmittance by 100 gives the percent
transmittance (%T), which varies between
100% (no absorption) and 0% (complete absorption). All methods of detection, whether
the human eye or a modern photoelectric transducer, measure
the transmittance of electromagnetic radiation.
Attenuation of radiation as it passes
through the sample
leads to a trans-
mittance of less than 1. As described, equation 10.1 does not distinguish be- tween the different ways in which
the attenuation of radiation occurs.
Besides absorption by the
analyte, several additional phenomena contribute to the net attenuation of radiation, including
reflection and absorption by the sample container, absorption by components of the sample
matrix other than the an- alyte, and the scattering of radiation. To compensate for this loss of the elec-
tromagnetic radiation’s power, we use a method blank (Figure 10.20b). The radiation’s power exiting from the method
blank is taken
to be P0.
An alternative method
for expressing the attenuation of electromagnetic
radiation is absorbance, A, which is defined as
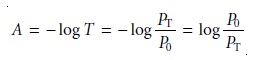 10.2
10.2
Absorbance is the more common unit for expressing
the attenuation of radiation because, as shown in the next section,
it is a linear function
of the analyte’s concentration.

Equation 10.1 has
an important consequence for atomic absorption. Because of the narrow
line width for atomic absorption, a continuum source
of radiation can- not
be used. Even with a high-quality monochromator, the effective bandwidth for a continuum source
is 100–1000 times
greater than that for an atomic absorption line. As a result,
little of the radiation from a continuum source is absorbed
(Po = PT), and
the measured absorbance is effectively zero.
For this reason,
atomic ab- sorption requires
a line source.
Related Topics