Chapter: 11th 12th std standard Class Physics sciense Higher secondary school College Notes
Semiconductors: Valence band, conduction band and forbidden energy gap
Semiconductors
It has been
observed that certain materials like germanium, silicon etc. have resistivity
between good conductors like copper and insulators like glass. These materials
are known as semiconductors. A material which has resistivity between
conductors and insulators is known as semiconductor. The resistivity of a
semiconductor lie approximately between 10-2 and 104 Ω m at room temperature. The resistance of a
semiconductor decreases with increase in temperature over a particular
temperature range. This behaviour is contrary to that of a metallic conductor
for which the resistance increases with increase in temperature.
The elements that are classified as semiconductors are Si, Ge, In, etc.
Germanium and silicon are most widely used as semiconductors.
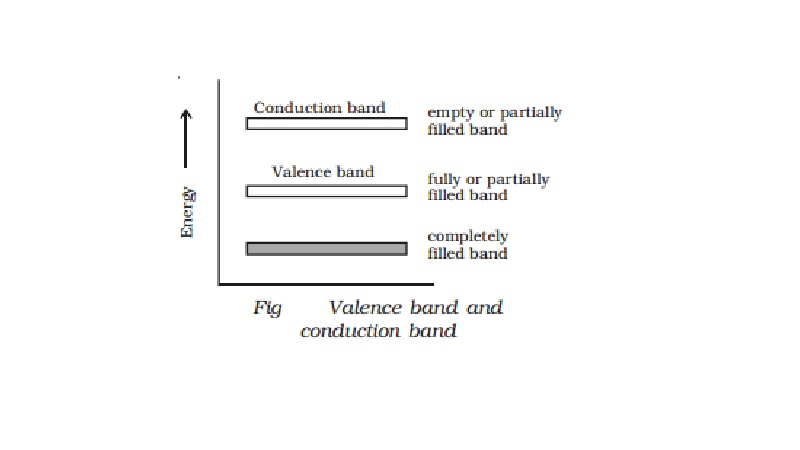
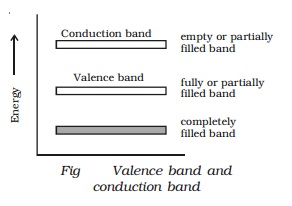
Valence band, conduction band and
forbidden energy gap
The
atoms of a solid are arranged in a regular repeated geometric pattern and the
electrons of the atom revolve around the nucleus in certain permitted energy
levels. The electrons in the inner shells are
trongly bound to the nucleus. A band which is occupied by the valence
electrons or a band having highest energy is defined as valence band (Fig). The
valence band may be partially or completely filled. This band can never be
empty.
In some materials, the valence
electrons are loosely attached to the nucleus. Even at room temperature, some
of the valence electrons can leave the valence band. These are called as free
electrons. They are responsible for conduction of current in a conductor and
are henceforth called as conduction electrons. The band occupied by these
electrons is called conduction band. This band may be an empty band or partially
filled band.
The separation between valence
band and conduction band is known as forbidden energy gap. If an electron is to
be transfered from valence band to conduction band, external energy is
required, which is equal to the forbidden energy gap.
Related Topics