Chapter: 11th 12th std standard Class Physics sciense Higher secondary school College Notes
Spontaneous and stimulated emission of Laser
Laser
The light emitted from an ordinary light source
is incoherent, because the radiation emitted from different atoms has no
definite phase relationship with each other. For interference of light,
coherent sources are essential. Two independent sources cannot act as coherent
sources. For experimental purposes, from a single source, two coherent sources
are obtained. In recent years, some sources have been developed, which are
highly coherent known as LASER. The word 'Laser' is an acronym for Light
Amplification by Stimulated Emission of Radiation. The difference between
ordinary light and laser beam is pictorially depicted in Fig .
Characteristics
of laser
The laser beam (i) is
monochromatic. (ii) is coherent, with the waves, all exactly in phase with one
another, (iii) does not diverge at all and (iv) is extremely intense
Spontaneous and stimulated emission
An atom may undergo
transition between two energy states E1 and E2, if it
emits or absorbs a photon of the appropriate energy E2 - E1
= hν.
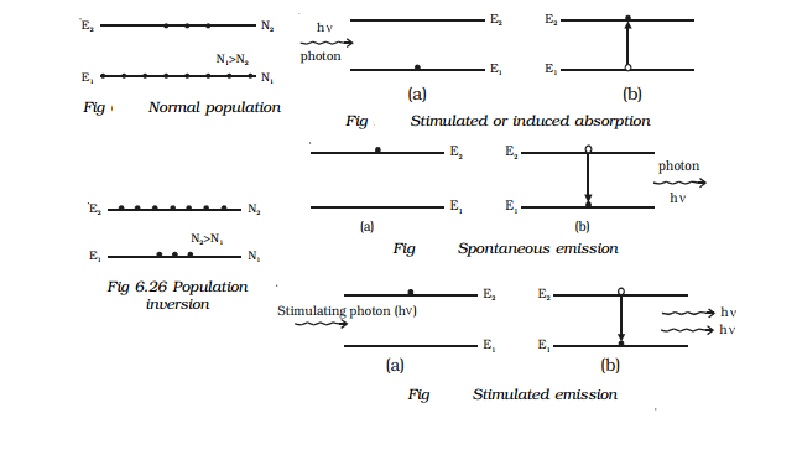
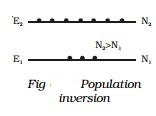
In
a system of thermal equilibrium, the number of atoms in the ground state (N1)
is greater than the number of atoms in the excited state (N2). This is called
normal population (Fig). Consider a sample of free atoms, some of which are in the ground state with
energy E1 and some in the excited energy state with energy E2.
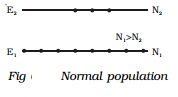
If photons of energy hν = E2-E1 are incident on
the sample, the photons can interact with the atoms in the ground state and are
taken to excited state. This is called stimulated or induced absorption (Fig
6.25). The process by which the atoms in the ground state is taken to the
excited state is known as pumping.
If the atoms are taken to the higher energy levels with the help of light, it is called optical pumping. If the atoms in the ground state are pumped to the excited state by means of external agency, the number of atoms in the excited state (N2) becomes greater than the number of atoms in the ground state (N1). This is E2 called population inversion (Fig ).
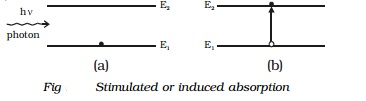
The life time of atoms in the excited E1 state is normally 10-8 second. Some of
the excited energy levels have
greater life times for atoms (10-3s).
Such energy levels are called as the metastable states. If the excited energy
level is an ordinary level, the excited atoms return to the lower (or) ground
energy state immediately without the help of any external agency. During this
transition (Fig), a photon of energy E2-E1 = hν is emitted. This is called spontaneous emission.
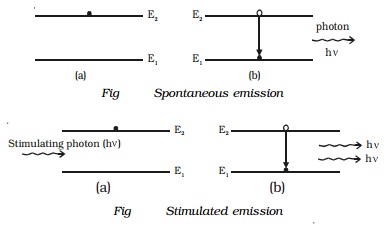
If the excited state is a metastable state, the
atoms stay for some time in these levels. The atoms in such metastable state
can be brought to the lower energy levels with the help of photons of energy hν = E2 - E1. During this process, a photon of
energy E2 - E1 = hν is emitted. This is known as stimulated
emission (or) induced emission (Fig 6.28). A photon produced by stimulated
emission is called secondary photon (or) stimulated photon. The secondary
photon is always in phase with the stimulating photon. These photons in turn
stimulate the emission further and the process continues to give a chain -
reaction. This is called laser action and by this action all the emitted
photons having same energy and same frequency are in phase with each other.
Hence, a highly monochromatic, perfectly coherent, intense radiation is
obtained in laser.
Conditions to achieve laser action
(i) There must be an inverted population i.e. more atoms
in the excited state than in the ground state.
(ii) The excited state must be a metastable state.
The emitted photons must stimulate further
emission. This is achieved by the use of the reflecting mirrors at the ends of
the system.
Related Topics