Chapter: 11th 12th std standard Class Physics sciense Higher secondary school College Notes
Insulators, semiconductors and conductors - forbidden energy gap
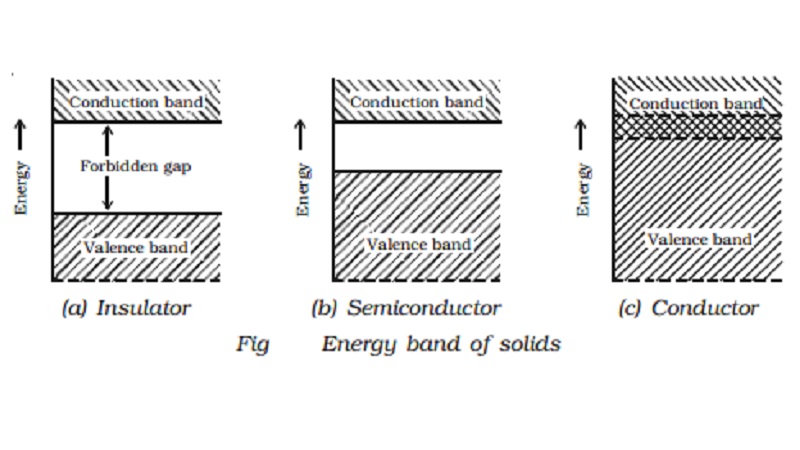
Insulators, semiconductors and conductors
Insulators
In an insulator, the forbidden
energy gap is very large (Fig a). In general, the forbidden energy gap is more
than 3eV and almost no electrons are available for conduction. Therefore, a
very large amount of energy must be supplied to a valence electron to enable it
to move to the conduction band. In the case of materials like glass, the
valence band is completely filled at 0 K. The energy gap between valence band
and conduction band is of the order of 10 eV. Even in the presence of high
electric field, the electrons cannot move from valence band to conduction band.
If the electron is supplied with high energy, it can jump across the forbidden
gap. When the temperature is increased, some electrons will move to the
conduction band. This is the reason, why certain materials, which are
insulators at room temperature become conductors at high temperature. The
resistivity of insulator approximately lies between 1011 and 1016
Ω m.
Semiconductors
In semiconductors (Fig b), the
forbidden gap is very small. Germanium and silicon are the best examples of
semiconductors. The forbidden gap energy is of the order of 0.7eV for Ge and
1.1eV for Si. There are no electrons in the conduction band. The valence band
is completely filled at 0 K. With a small amount of energy that is supplied,
the electrons can easily jump from the valence band to the conduction band. For
example, if the temperature is raised, the forbidden gap is decreased and some
electrons are liberated into the conduction band. The conductivity of a
semiconductor is of the order of 102 mho m-1.
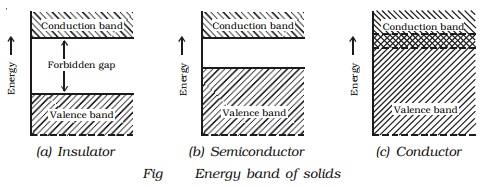
Conductors
In conductors, there is no
forbidden gap available, the valence and conduction band overlap each other
(Figc). The electrons from valence band freely enter into the conduction band.
Due to the overlapping of the valence and conduction bands, a very low
potential difference can cause the continuous flow of current.
Related Topics