Chapter: Genetics and Molecular Biology: Protein Synthesis
Protein Elongation - Protein Synthesis
Protein Elongation
Although
it would appear that the charged tRNAs could diffuse into the ribosome and bind
to the codons of mRNA, they are in fact carried into the binding sites on a
protein. The protein that serves this function during elongation was originally
called Tu (unstable) but is now some-times called EF1 (elongation
factor 1). A rather complex cycle is used for carrying the charged tRNAs to the
ribosome A site (Fig. 7.13). First, GTP binds to EF1, then an
aminoacyl tRNA binds, and this complex enters the ribosome A site containing a
complementary codon. There GTP is hydrolyzed to GDP, and EF1-GDP is
ejected from the ribosome. The completion of the cycle takes place in solution.
GDP is displaced from EF1-GDP by EF2, which in turn is
displaced by GTP. The binding of GDP to EF1 is tight; Kd
equals approximately 3 × 10-9
M. This plus the fact that EF1 binds to filters permits a simple
filter-binding assay to be used to quantitate the protein. Originally the very
tight binding for GDP generated confusion because the commercial preparation of
GTP con-tained minor amounts of contaminating GDP.
Both the
initiation factor IF2 and the elongation factor EF1 carry
charged tRNA molecules into the ribosome. Not surprisingly, they possess
considerable amino acid sequence homology. Additionally, cells which
incorporate selenocysteine into the one or two proteins contain-ing this amino
acid use yet a third factor. This carries the charged tRNA into the ribosome,
and, as expected, also possesses significant homology with the other two
factors. All three of the proteins are members of the broad and important class
of proteins known as G proteins as they bind GTP. In eukaryotic cells the G
proteins most often are part of signal transduction pathways from receptor proteins bound in the membrane to
gene regulation or other intracellular points of regulation.
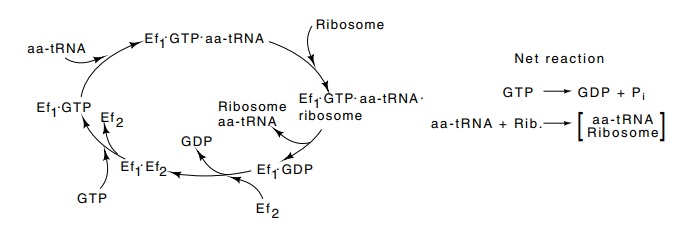
Figure
7.13 The cycle by which EF1carries
GTP and aa-tRNA to the ribosomeduring protein synthesis.
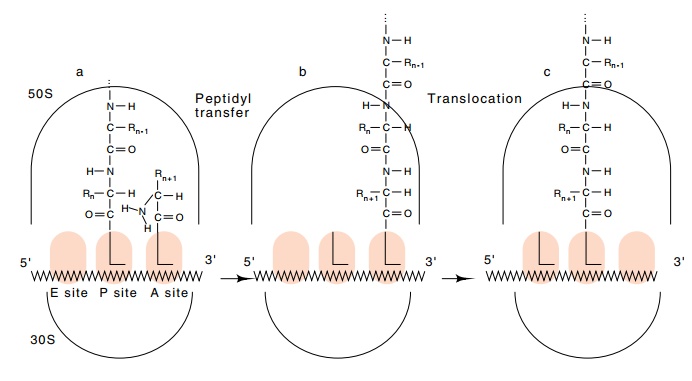
Figure
7.14 The process of protein synthesis.
(a) The polypeptide occupies theP site, and the incoming aa-tRNA occupies the A
site. (b) After formation of the next peptide bond, the polypeptide occupies
the A site and the tRNA in the P site is not acylated. (c) The ribosome
following translocation. The elongated polypeptide now occupies the P site, the
A site is empty, awaiting arrival of EF1 with another aa-tRNA, and the
discharged tRNA occupies the E site.
Related Topics