Kinetic Theory of Gases | Physics - Solved Example Problems for Mean Free Path | 11th Physics : UNIT 9 : Kinetic Theory of Gases
Chapter: 11th Physics : UNIT 9 : Kinetic Theory of Gases
Solved Example Problems for Mean Free Path
Physics : Kinetic Theory of Gases - Solved Example Problems for Mean Free Path
EXAMPLE 9.6
An oxygen molecule is travelling in air at 300 K and 1 atm, and the diameter of oxygen molecule is 1.2 × 10−10m. Calculate the mean free path of oxygen molecule.
Solution
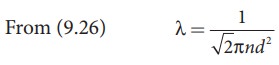
We have to find the number density n By using ideal gas law
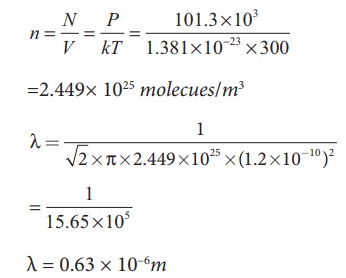
Tags : Kinetic Theory of Gases | Physics , 11th Physics : UNIT 9 : Kinetic Theory of Gases
Study Material, Lecturing Notes, Assignment, Reference, Wiki description explanation, brief detail
11th Physics : UNIT 9 : Kinetic Theory of Gases : Solved Example Problems for Mean Free Path | Kinetic Theory of Gases | Physics
Related Topics
11th Physics : UNIT 9 : Kinetic Theory of Gases