Chapter: 11th 12th std standard Class Physics sciense Higher secondary school College Notes
Electrons and holes in semiconductors
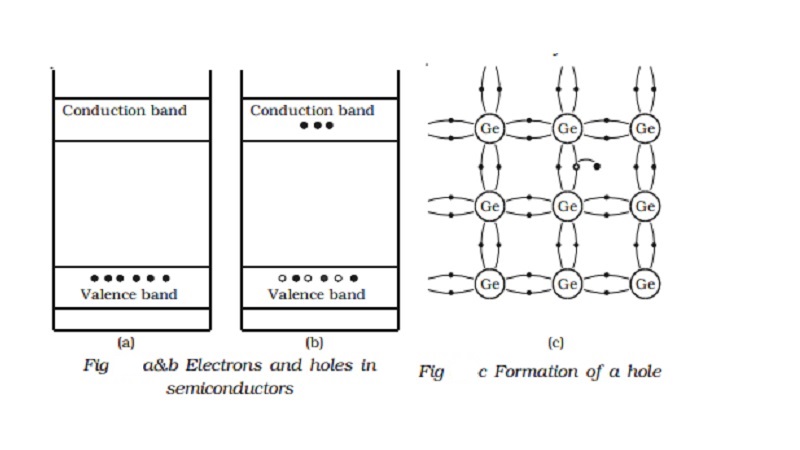
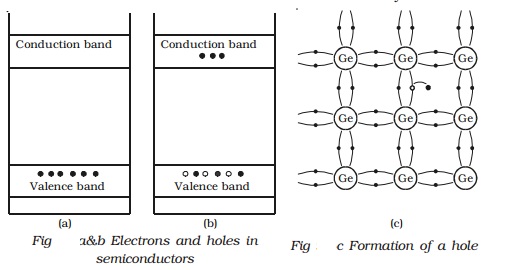
Electrons and holes in
semiconductors
Fig b shows the energy band
diagram of an intrinsic semiconductor (pure semiconductor). Fig a and Fig b
represent charge carriers at absolute zero temperature and at room temperature
respectively.
The electrons in an intrinsic
semiconductor, which move in to the conduction band at high temperatures are
called as intrinsic carriers. In the valence band, a vacancy is created at the
place where the electron was present, before it had moved in to the conduction
band. This vacancy is called hole. Fig c helps in understanding the creation of
a hole. Consider the case of pure germanium crystal. It has four electrons in
its outer or valence orbit. These electrons are known as valence electrons.
When two atoms of germanium are brought close to each other, a covalent bond is
formed between the atoms. If some additional energy is received, one of the
electrons contributing to a covalent bond breaks and it is free to move in the
crystal lattice.
While coming out of the bond, a
hole is said to be created at its place, which is usually represented by a open
circle. An electron from the neighbouring atom can break the covalent bond and
can occupy this hole, creating a hole at another place. Since an electron has a
unit negative charge, the hole is associated with a unit positive charge. The
importance of hole is that, it may serve as a carrier of electricity in the same
manner as the free electron, but in the opposite direction.
Related Topics