Chapter: 11th Chemistry : UNIT 13 : Hydrocarbons
Preparation of benzene
Sources of aromatic compound:
Benzene and other aromatic compound are obtained from coal tar and petroleum
It can also be prepared in laboratory using some simple aliphatic compounds
Preparation of benzene
(i) industrial preparation of benzene from coal tar :
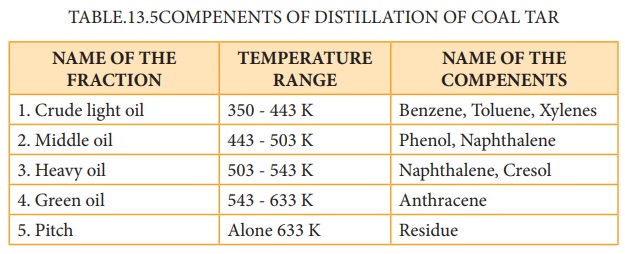
Coal tar is a viscous liquid obtained by the pyrolysis of coal. During fractional distillation, coal tar is heated and distills away its volatile compounds namely benzene, toluene, xylene in the temperature range of 350 to 443 K. These vapours are collected at the upper part of the fractionating column (Table 13.5.)
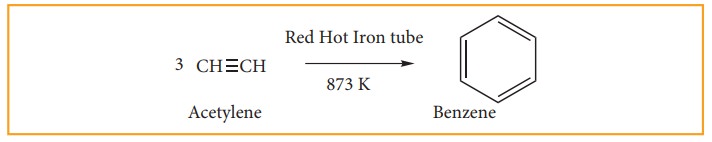
(ii) from acetylene.
Acetylene on passing through a red –hot tube trimerises to give benzene. We have already studied this concept in polymerization of alkynes.
(iii) Laboratory Methods Of Preparing Benzene And Toluene
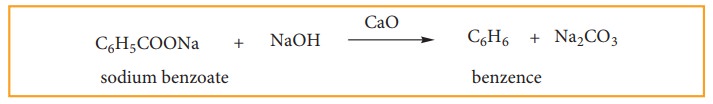
(a) Decarboxylaation Of Aromatic Acid.
When sodium benzoate in heated with sodalime, benzene vapours distil over.
(b) Preparation Of Benzene From Phenol
When phenol vapours are passed over zinc dust, then it is reduced to benzene.

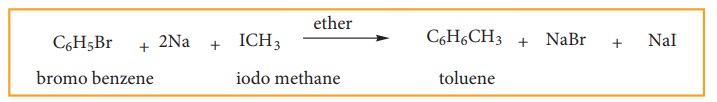
(c) Wurtz – Fittig Reaction:
When a solution of bromo benzene and iodo methane in dry ether is treated with metallic sodium, toluene is formed.
(d) Friedel Craft’s Reaction:
When benzene is treated with methyl chloride in the presence of anhydrous aluminium chloride, toluene is formed.

Related Topics