Chapter: 11th Chemistry : UNIT 13 : Hydrocarbons
Preparation of alkanes
Preparation of alkanes:
Alkanes are not laboratory curiosities but they are extremely important naturally occurring compounds. Natural gas and petroleum (crude oil) are the most important natural sources. However, it can be prepared by the following methods.
1. Preparation of alkanes from catalytic reduction of unsaturated hydro-carbons.
When a mixture hydrogen gas with alkene or alkyne gas is passed over a catalysts such as platinum or palladium at room temperature, an alkane is produced. This process of addition of H2 to unsaturated compounds is known as hydrogenationThe above process can be catalysed by nickel at 298K. This reaction is known as Sabatier-Sendersens reaction
for example:
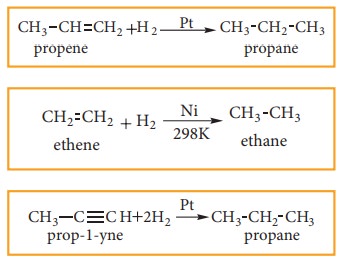
2. Preparation of alkanes from carboxylic acids:
i) Decarboxylation of sodium salt of carboxylic acid
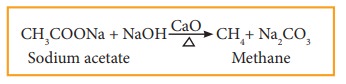
When a mixture of sodium salt of carboxylic acid and soda lime (sodium hydroxide + calcium oxide) is heated, alkane is formed. The alkane formed has one carbon atom less than carboxylic acid. This process of eliminating carboxylic group is known as decarboxylation.
for example:
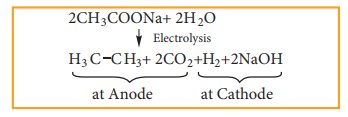
ii) Kolbe’s Electrolytic method
When sodium or potassium salt of carboxylic acid is electrolyzed, a higher alkane is formed. The decarboxylative dimerization of two carboxylic acid occurs. This method is suitable for preparing symmetrical alkanes(R-R).
3.Preparation of alkanes using alkyl halides (or) halo alkanes
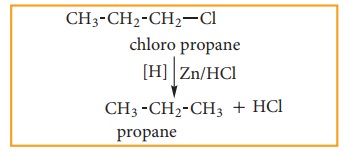
i) By reduction with nascent hydrogen
Except alkyl fluorides, other alkyl halides can be converted to alkanes by reduction with nascent hydrogen. The hydrogen for reduction may be obtained by using any of the following reducing agents: Zn+HCl, Zn+CH3COOH, Zn-Cu couple in ethanol, LiAlH4 etc.,
for example:
ii) Wurtz reaction
When a solution of halo alkanes in dry ether is treated with sodium metal, higher alkanes are produced. This reaction is used to prepare higher alkanes with even number of carbon atoms.
for example:
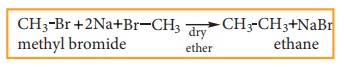
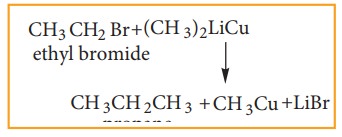
iii) Corey- House Mechanism
An alkyl halide and lithium di alkyl cuprate are reacted to give higher alkane.
for example:
4) Preparation of Alkanes from Gri-gnard reagents
Halo alkanes reacts with magnesium in the presence of dry ethers to give alkyl magnesium halide which is known as Grignard reagents. Here the alkyl group is directly attached to the magnesium metal make it to behave as carbanion. So, any compound with easily replaceable hydrogen reacts with Grignard reagent to give corresponding alkanes.
for example:
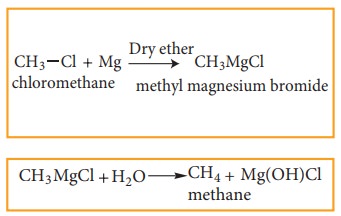
Related Topics