Chapter: 11th 12th std standard Class Physics sciense Higher secondary school College Notes
Perfect black body
Perfect black body
A perfect black
body is the one which absorbs completely heat radiations of all wavelengths
which fall on it and emits heat radiations of all wavelengths when heated.
Since a perfect black body neither reflects nor transmits any radiation, the
absorptive power of a perfectly black body is unity.
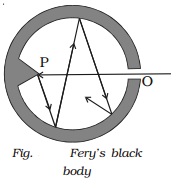
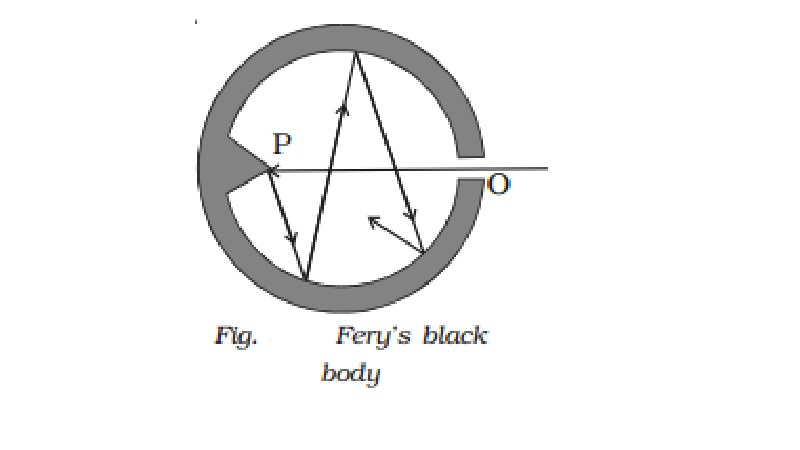
1 Fery?s black body
Fery?s black body consists of a
double walled hollow sphere having a small opening O on one side and a conical
projection P just opposite to it (Fig.). Its inner surface is coated with lamp
black. Any radiation entering the body through the opening O suffers multiple
reflections at its innerwall and about 97% of it is absorbed by lamp black at
each reflection. Therefore, after a few reflections almost entire radiation is
absorbed. The projection helps in avoiding any direct reflections which even
otherwise is not possible because of the small opening O. When this body is
placed in a bath at fixed temperature, the heat radiations come out of the
hole. The opening O thus acts as a black body radiator.
2.Prevost?s theory of heat exchanges
Prevost applied the idea of
thermal equilibrium to radiation. According to him the rate at which a body
radiates or absorbs heat depends on the nature of its surface, its temperature
and the temperature of the surroundings. The total amount of heat radiated by a
body increases as its temperature rises. A body at a higher temperature
radiates more heat energy to the surroundings than it receives from the
surroundings. That is why we feel warm when we stand before the furnace.
Similarly a body at a lower
temperature receives more heat energy than it loses to the surroundings. That
is why we feel cold when we stand before an ice block.
Thus the rise or fall of
temperature is due to the exchange of heat radiation. When the temperature of
the body is the same as that of surroundings, the exchanges of heat do not
stop. In such a case, the amount of heat energy radiated by the body is equal
to the amount of heat energy absorbed by it.
A body will stop emitting
radiation only when it is at absolute zero. (i.e) 0 K or ?273o C. At
this temperature the kinetic energy of the molecule is zero.
Therefore, Prevost theory states
that all bodies emit thermal radiation at all temperatures above absolute zero,
irrespective of the nature of the surroundings.
3 Kirchoff?s Law
According to this law, the ratio of emissive power to the
absorptive power corresponding to a
particular wavelength and at a given temperature is always a constant for all
bodies. This constant is equal to the emissive power of a perfectly black body at the same temperature and the
same wavelength. Thus, if eλ is the emissive power of a body corresponding to a wavelength λ at any given temperature, aλ is the absorptive power of the
body corresponding to the same wavelength at the same temperature and Eλ is the emissive power of a
perfectly black body corresponding to the same wavelength and the same
temperature, then according to Kirchoff?s law
eλ/ aλ = constant = Eλ
From the above equation it is
evident that if aλ is large, then eλ will also be large (i.e) if a body absorbs radiation of certain
wavelength strongly then it will also strongly emit the radiation of same
wavelength. In other words, good absorbers of heat are good emitters also.
Applications of Kirchoff?s law
(i)The silvered surface of a thermos flask is a bad
absorber as well as a bad radiator. Hence, ice inside the flask does not melt
quickly and hot liquids inside the flask do not cool quickly.
(ii)
Sodium vapours on heating, emit two bright yellow lines. These
are called D1 and D2 lines of sodium. When
continuous white light from carbon arc passes through sodium vapour at low
temperature, the
continuous spectrum is absorbed
at two places corresponding to the wavelengths of D1 and D2
lines and appear as dark lines. This is in accordance with Kirchoff?s law.
4 Wien?s displacement law
Wien?s displacement law states that the wavelength of the radiation
corresponding to the maximum energy (λm) decreases as the temperature T
of the body increases.
(i.e) λm T = b where b is called Wien?s constant. Its value is 2.898 ? 10-3 m
K
5 Stefan?s law
Stefan?s law states that the total amount of heat energy radiated
per second per unit area of a perfect
black body is directly proportional to the fourth power of its absolute temperature.
(i.e) E α T4 or E = σT4
where σ is called the Stefan?s constant. Its value is 5.67 ?
10-8 W m-2 K-4.
It is also called Stefan -
Boltzmann law, as Boltzmann gave a theoretical proof of the result given by
Stefan.
6 Newton?s law of cooling
Newton?s law of cooling states
that the rate of cooling of a body is
directly proportional to the temperature
difference between the body and the surroundings.
The law holds good only for a
small difference of temperature. Loss of heat by radiation depends on the
nature of the surface and the area of the exposed surface.
Related Topics