Chapter: 11th 12th std standard Class Physics sciense Higher secondary school College Notes
Experiments to demonstrate surface tension
Surface tension of a liquid
Surface tension is the property of the free surface of a liquid at
rest to behave like a stretched membrane in order to acquire minimum surface
area.
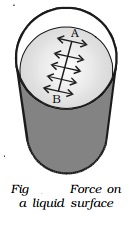
Imagine a line AB in the free surface of a liquid at rest (Fig.).
The force of surface tension is measured as the force acting per unit length on
either side of this imaginary line AB. The force is perpendicular to the line
and tangential to the liquid surface. If F is the force acting on the length l
of the line AB, then surface tension is given by T=F/l.
Surface tension is defined as the force per unit length acting
perpendicular on an imaginary line drawn on the liquid surface, tending to pull
the surface apart along the line. Its unit is N m-1 and dimensional
formula is MT-2.
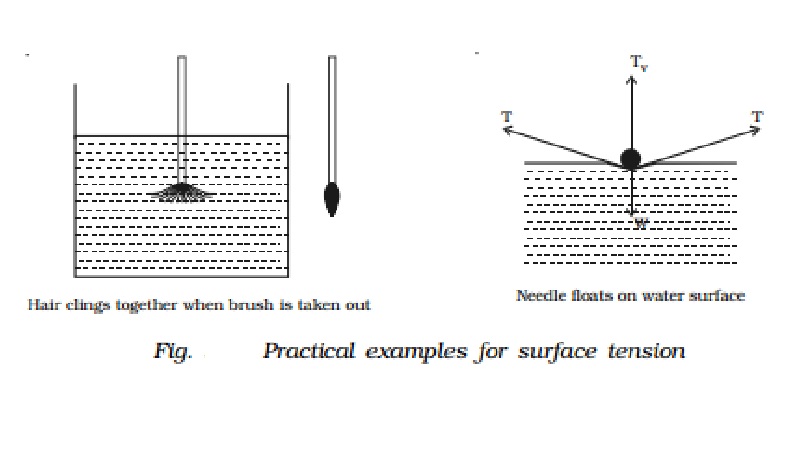
Experiments to demonstrate surface tension

(i)
When a painting brush is dipped into water, its
hair gets separated from each other. When the brush is taken out of water, it
is observed that its hair will cling together. This is because the free surface
of water films tries to contract due to surface tension.
(ii)
When a sewing needle is gently placed on water
surface, it floats. The water surface below the needle gets depressed slightly.
The force of surface tension acts tangentially. The vertical component of the
force of surface tension balances the weight of the needle.
Related Topics