Chapter: The Massage Connection ANATOMY AND PHYSIOLOGY : Cardiovascular System
The Heart: Structure and Function
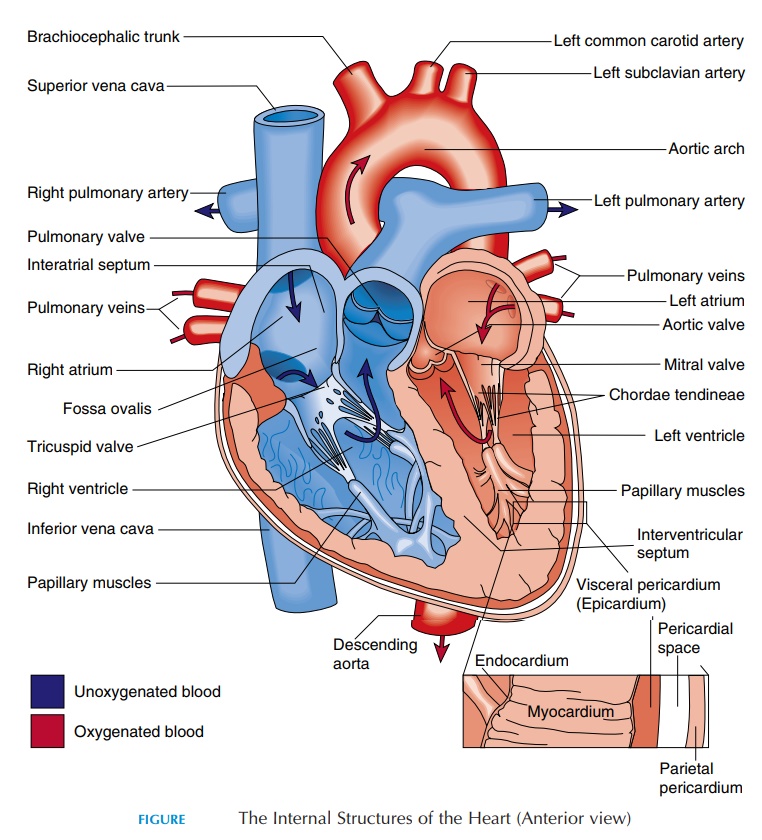
The Heart
The heart is an organ that is classically described to be the size of a clenched fist. It is located anteriorly, just behind the sternum. A major portion of the heart is situated towards the left side of the body.
The heart rests on the diaphragm, wedged between the two lungs (pleural cavities) in the mediastinum of the thorax. The mediastinum is the portion of the tho- racic cavity located between the pleural cavities, and it houses the heart, thymus, trachea, and esophagus.
The Pericardium
The heart is surrounded by a fluid-filled cavity known as the pericardial cavity. This cavity is lined by a membrane—the pericardium. The pericardium is a serous membrane, reinforced by dense connec-tive tissue. The dense connective tissue is often re-ferred to as the fibrous pericardium, and the serous membrane as the serous pericardium. The rela-tionship of the heart to the pericardium (Figure 8.7C) is like a fist pushed into a partially filled bal-loon. In the latter, two layers separated by air will cover the fist, with one of the layers in close contact with the fist. In the case of the heart, the layer close to the heart is called the visceral pericardium or epicardium.The other layer is known as the pari-etal pericardium. Instead of air, this space is filledwith the pericardial fluid. The point where the wrist would have entered the balloon is equivalent to the region where the large blood vessels enter and leave the heart. Although located superiorly, this region is known as the base of the heart. The inferior pointed tip of the heart (located to the left) is known as the apex.
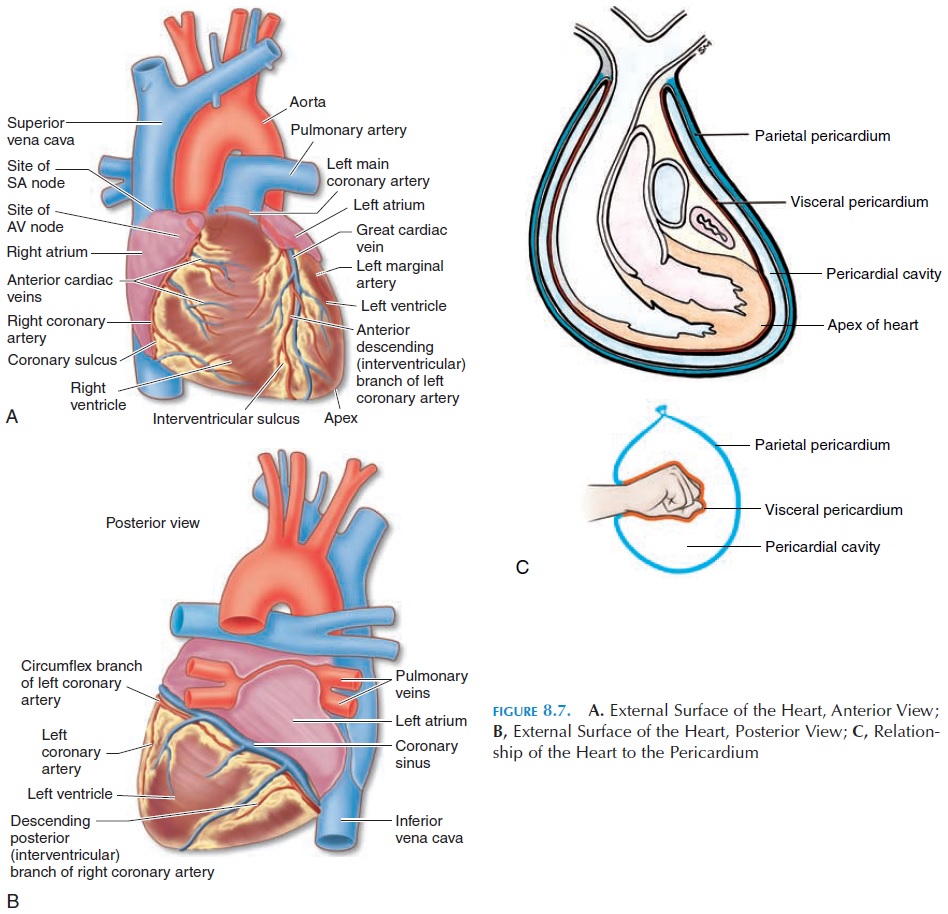
The function of the pericardium is to prevent the heart from expanding too much and enlarging ab-normally as could happen if too much blood re-turned to the heart. In addition, it helps hold the heart in position as the fibrous pericardium fuses with the diaphragm inferiorly and the outer wall of the blood vessels superiorly. The pericardial fluid, which is about 10–20 mL (1.1–2.1 qt), is a lubricant that reduces friction between surfaces as the heart beats.
Chambers of the Heart
The human heart has four chambers—the rightatrium, left atrium, right ventricle, and left ventri-cle. The atriareceiveblood, while the ventricleseject/expel the blood. The right atrium communicateswith the right ventricle, the left atrium with the left ventricle. In normal individuals, there is no direct communication between the chambers on the right side and the left side.
The location and boundaries of the four chambers can be identified on the surface of the heart. A deep groove filled with fat, the coronary sulcus, marks the boundary between the atria and ventricles (Figure 8.7). The coronary arteries and veins (blood vessels that supply the walls of the heart) run in this groove. Other depressions located anteriorly and posteriorly, the anterior and posterior interventricular sulcus, mark the boundary between the two ventricles (Fig-ure 8.7).
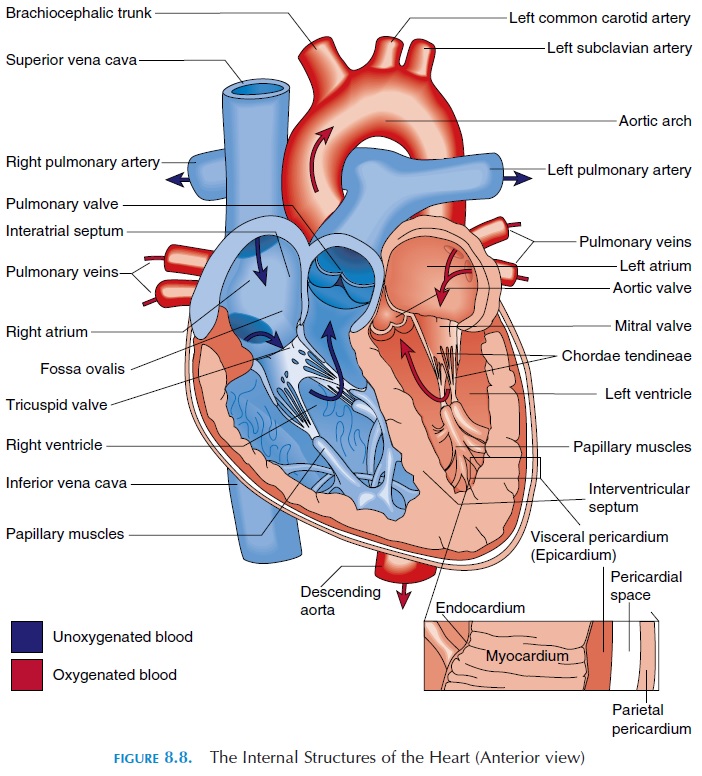
The Right Atria
The two atria (see Figure 8.8) are separated by the interatrial septum. In the fetus, the two atria com-municate with each other through an oval opening, the foramen ovale, shunting the blood from right to left because the lungs do not function and the pla-centa takes the place of lungs. This opening closes at birth when the baby takes its first breath and re-mains as an oval depression (fossa ovalis). The right atrium communicates with two large veins—the su-perior vena cava and the inferior vena cava.Theseveins drain blood from the upper and lower part of the body, respectively. The superior vena cava deliv-ers blood to the atrium from the head, neck, upper limbs, and chest, and the inferior vena cava carries blood from the rest of the trunk, the viscera, and the lower limbs. Blood from the heart walls is drained by coronary veins into a larger coronary sinus that opens into the right atrium. The inside wall of the right atrium is smooth, but has prominent muscular ridges.
Atrioventricular Valves
The atrium and ventricle on each side are separated by one-way valves known as atrioventricular valves(AV valves). The one on the right is the tricuspid valve, and the one on the left is the mitral or bicus-pid valve. These valves have three and two cusps, re-spectively. Cusps are thin membranes or folds of fi-brous tissue that are attached to the inner walls of the ventricles. When the cusps come together, blood can-not flow from the atrium into the ventricle. When the ventricle relaxes, the cusps are pulled apart, allowing blood to flow. The cusps are attached to the inner wall of the ventricle by thin, but tough, stringlike connective tissue known as the chordae tendineae.These are then connected to prominent muscular projections known as papillary muscles.
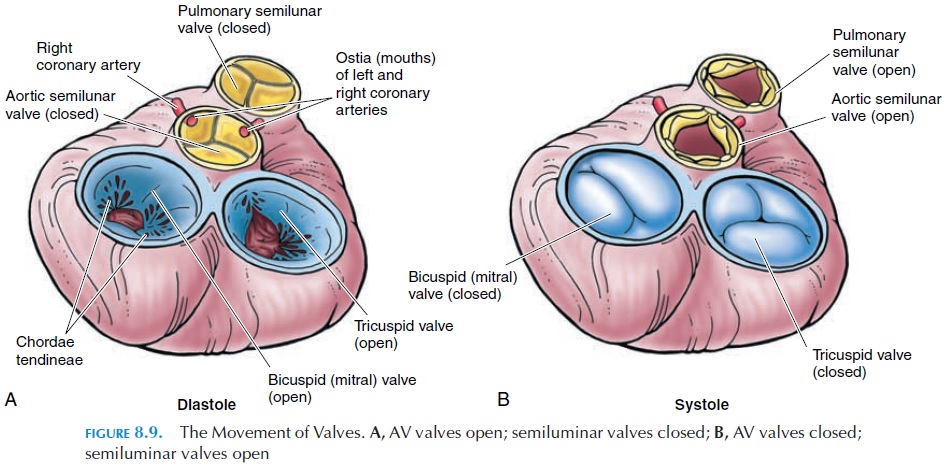
The valves open or close, according to the differ-ence in pressure in the two chambers (see Figure 8.9). When the ventricle relaxes, the pressure drops below that of the atria and the valves open. The cusps point toward the ventricle and the chordae tendineae are relaxed. When the ventricle contracts, the pres-sure increases and the edges of the cusps are brought together, closing the opening between the two cham-bers. At the same time, the papillary muscles con-tract, pulling the chordae tendineae taut and pre-venting the cusps from everting into the atrium.
The Right Ventricle
The right atrium connects with the right ventricle. The right ventricle is separated from the left ventricle by the interventricular septum. The superior end of the ven-tricle tapers and opens into a large blood vessel—the pulmonary trunk.
The opening is guarded by a valve known as the pulmonary semilunar valve. This one-way valve has three moon-shaped cusps that open when the ventricle contracts and close when it relaxes, allow-ing blood to pass from the ventricle to the pulmonary trunk. The pulmonary trunk branches into the right and left pulmonary arteries soon after it leaves the ventricle. These arteries branch repeatedly after they enter the lungs, finally forming capillaries where gas exchange takes place. The oxygenated blood from the pulmonary capillaries flows into the venules and then into the four pulmonary veins.
The Left Atrium
The four pulmonary veins (two left and two right) open into the posterior wall of the left atrium. Blood from the left atrium flows into the left ventricle. The opening between the atrium and ventricle, as with the right side of the heart, is guarded by the atrioventric-ular valve. This valve has only two cusps instead of three and is referred to as the mitral or bicuspidvalve (mitre,a bishop’s headpiece). This valve opens when the ventricles relax and closes when the ventri-cles contract, allowing blood to flow one way, from the atrium to the ventricle. These valves, similar to those on the left, are attached to the walls of the ven-tricle by chordae tendineae and papillary muscles.
The Left Ventricle
The walls of the left ventricle are much thicker and more muscular than that of the right as they have to withstand a much higher pressure of blood. The higher pressure is needed here to push the blood through the systemic circulation. The wall of the right ventricle is much thinner as it only has to push blood into the lungs, which offer much less resis-tance. Unlike the various organs in the systemic cir-culation, the lungs are located close to the heart and not much pressure is required for blood to flow through them.
The blood from the ventricle is pumped into the largest of the blood vessels—the aorta. The opening into the aorta is guarded by the aortic semilunarvalve, similar to that on the right. This one-way valveopens when the ventricle contracts and closes as it re-laxes, allowing blood to pass from the ventricle to the aorta.
The Heart Wall
If the heart wall is cut, three distinct layers can be iden-tified (see Figure 8.8). The outermost layer is the epi-cardium or visceral pericardium already described.Deep to it is the myocardium or muscular wall of the heart. The myocardium contains cardiac muscle tissue, blood vessels, and nerves. The muscles are arranged in such a way that, when the muscle contracts, blood is squeezed out of the heart into the blood vessels. The thickness of the heart wall varies according to the pres-sure it has to withstand. The atria are thin walled be-cause they are exposed to low venous pressure. The wall of the right ventricle is also thin as a result of the low resistance offered by the lungs. The left ventricle has the thickest wall because it works the hardest.
The innermost layer of the heart is the endo-cardium. This layer also lines the heart valves. Theendocardium consists of simple squamous epithe-lium that is continuous with the endothelium of the blood vessels.
Connective Tissue and the Fibrous Skeleton
For greatest efficiency, the atrium has to contract a little before the ventricle to allow time for the blood from the atrium to reach the ventricle before it con-tracts. This is accomplished because the muscles around the atrium and the ventricles are separated by a fibrous tissue ring. This connective tissue skeleton surrounds the valves, the aorta, and the pulmonary trunk, isolating the atrial and ventricular muscle. Al-though there is no direct connection between the muscles of the atrium and the ventricle, specialized tissue known as the conducting system pierce through the fibrous ring and convey impulses gener-ated in the atrium to the ventricle.
In addition, the connective tissue fibers provide physical support for the cardiac muscle fibers, nerves, and blood vessels; help distribute the force of contraction and strength; prevent excessive stretch-ing of the heart, and provide elasticity to help the heart return to its original size.
Cardiac Muscle
Cardiac muscle (see Figure 4.18) is striated similar to skeletal muscle. The contraction physiology is the sim-ilar to that of skeletal muscle; however, the structure of cardiac muscle is slightly different. Cardiac muscle fiber is shorter and broader than skeletal muscle fiber. It is branched, with a centrally located single nucleus. Individual muscle cells are interconnected by interca-lated disks. These disks help convey the force of con-traction and impulses from one cell to another. As a re-sult, if an impulse is initiated in one of the cardiac muscle fibers, it is conveyed to all the muscles, and the heart contracts as a functional syncytium (i.e., as if one big muscle). Because the atria are separated from the ventricles by the fibrous tissue skeleton, the two atria contract as one functional syncytium, pushing the blood into the ventricle; the ventricles contract as another functional syncytium, pushing the blood into the arteries (pulmonary trunk and aorta).
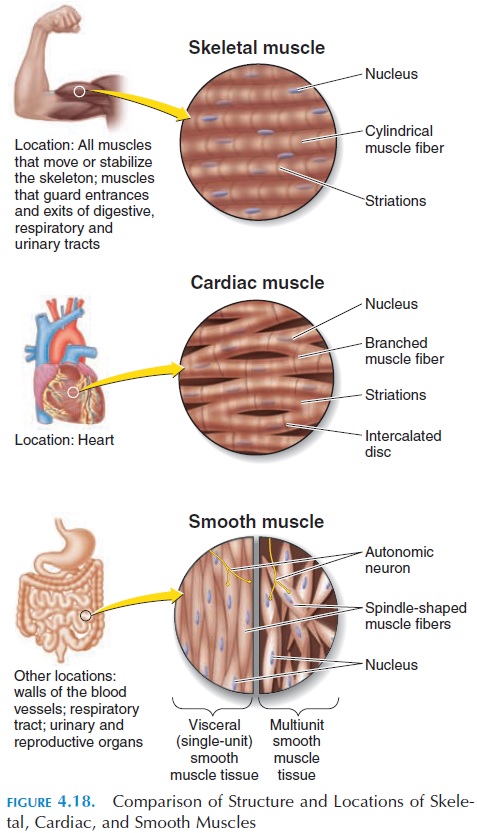
The calcium in skeletal muscle is only derived from the sarcoplasmic reticulum; in cardiac muscle, calcium is also obtained from the interstitial fluid surrounding the muscle. The contractility of the car-diac muscle is affected by hormones and ionic changes in the blood.
Action Potential in Cardiac Muscle
Another difference between cardiac muscle and skeletal muscle is the action potential—the ventricu-lar muscle is about 30 times as long as the action po-tential in the skeletal muscle and lasts for about 250–300 milliseconds (see Figure 8.11A).
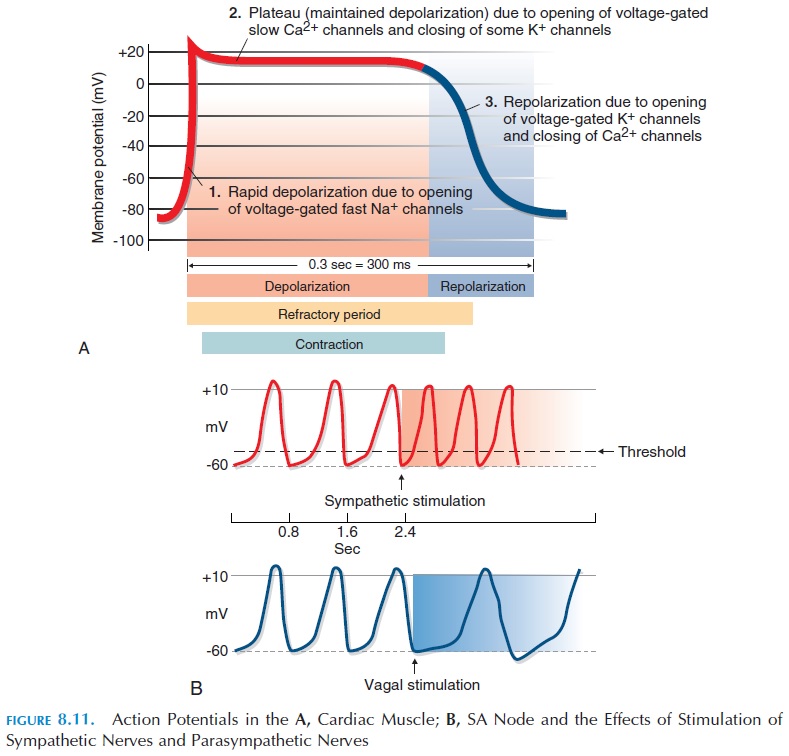
The resting membrane potential of the muscle fiber is about -90 mV. When the cardiac muscle fiber is stim-ulated, voltage-gated fast sodium channels present in the sarcolemma open, allowing sodium to rush into the cell (at rest, there is more sodium ions outside the cell). This produces depolarization. Soon after, the sodium channels close and voltage-gated calcium channels open, allowing positively charged calcium ions to enter. At the same time, there is a slow leak of potassium ions out of the cell. This results in a plateau phase that is responsible for prolonging the duration of the action potential. Note that this phase is absent in the action potential of skeletal muscles. Following the plateau phase, voltage-gated potassium channels open wider, allowing positively charged potassium to leak out and produce repolarization. This brings the membrane potential back to its resting state.
Excitation-Contraction Coupling
The action potential in the cardiac muscle leads to con-traction. The mechanism is similar to that in skeletal muscle. When the cell depolarizes, cal-cium levels rise inside the cytoplasm. The calcium at-taches to troponin, which, in turn, triggers actin and myosin to slide past each other, causing a contraction.
Refractory Period
The time interval in which a second contraction cannot be triggered is referred to as the refractory period of that muscle (see Figure 8.11). The cardiac muscle has a long refractory period compared with skeletal muscle because of the plateau phase of the action potential.
The longer duration of cardiac action potential (i.e., refractory period) is beneficial. For instance, if another impulse reaches the muscle during the first action potential, it cannot produce another contrac-tion and results in sustained (tetanic) contraction of the muscle. In the heart, if tetanic contractions were possible, it would have serious consequences because filling of blood in the ventricular chamber would be jeopardized and blood will not be ejected to supply the brain and other parts of the body.
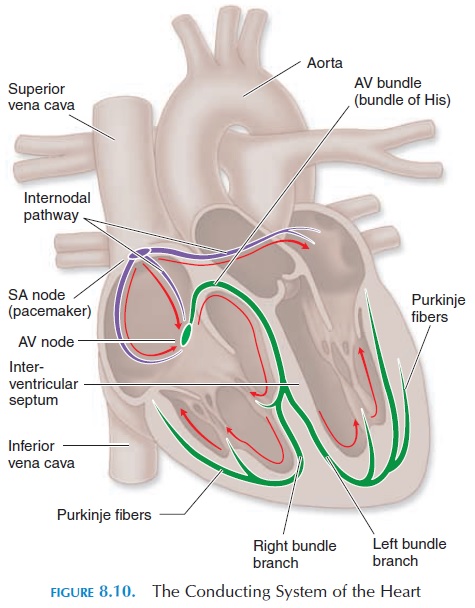
Conducting Tissue of the Heart
Unlike skeletal muscle, cardiac muscle has the capac-ity to contract on its own in the absence of stimulation by nerves or hormones. This property is referred to asautomaticity, which is a result of specialized cardiacmuscle tissue (autorhythmic cells) in the heart known as the conducting system. The conducting system network initiates and conducts and distributes electri-cal impulses.

The components of the conducting system are shown in Figure 8.10. It includes the sinoatrial (SA) node, located in the wall of the right atrium; the atrio-ventricular(AV) node, located at the junction of theatrium and ventricle; the conducting cells of the in-ternodal pathway that interconnect the SA and theAV nodes and convey impulses to the muscles of the atrium; the atrioventricular (AV) bundle (bundle of His); the right and left bundle branches that convey impulses towards the right and left ventricle; and the Purkinje fibers that distribute the impulses to theventricular muscle fibers.
Generation of Rhythmic Impulse
The action potential of conducting tissue is different from that of skeletal muscle or the ventricular muscle described above. The cells in the conducting system are smaller than other muscle fibers of the myo-cardium and, unlike the others, cannot maintain a stable, resting membrane potential. Every time these cells reach their resting potential after depolarization, there is a slow leak of positive ions into the cell, raising the potential toward threshold and triggering another action poten-tial (see Figure 8.11B). The rate at which action poten-tials are triggered is fastest in the SA node, about 80–100 times/minute. The other parts of the conducting system also have an unstable resting potential, how- ever, action potentials are triggered here at a slower rate.
For example, the AV node can generate action po-tentials at the rate of 40–60/minute; the rest of the con-ducting system generates at an even slower rate. Be-cause the SA node generates action potentials at the fastest rate, the heart rate is normally determined by its pace. Hence, the SA node is known as the pacemaker.
Conduction of Impulses
Impulses generated by the pacemaker are conducted rapidly to the AV node along the internodal pathway. At the same time, the impulses are conducted to the atrial muscle, and the two atrium contract as a functional syncytium. The impulses do not travel to the ventricle directly from the atrium as a result of the presence of the nonconducting connective tissue of the fibrous skeleton, which separates the atrium from the ventricle.
At the AV node, the conduction of impulses that have reached it from the SA node is slowed down. This is advantageous, as it gives the atrium enough time to contract and propel the blood into the ventri-cles. The cells of the AV node can conduct impulses at a maximal rate of about 230/minute. As each impulse can produce a ventricular contraction, this is the maximal heart rate of an individual.
The connection between the AV node and the AV bundle is the only route through which impulses can pass from the atrium to the ventricle. The AV bundle splits into right and left bundle branches located in the interventricular septum.
From here, impulses travel through the Purkinje fibers that rapidly con-duct impulses to all ventricular muscle fibers. Ven-tricular contraction proceeds from the apex in a wave and spreads toward the base, squeezing the blood to-ward the base where the large vessels are located.
An Overview of the Electrocardiogram
By placing electrodes on the surface of the chest wall, the electrical changes that occur in the heart can be recorded. The electrocardiogram (ECG) (see Figure 8.12) is the recording of these electrical activities. The general direction of conduction of the electrical impulse is from the SA node through the atria to the AV node, then along the AV bundle in the interven-tricular septum to the apex, and then along the ven-tricular wall toward the base of the heart (Figure 8.10). When the impulse is conducted, the inside of the myocardial cell becomes positive and the outside becomes negative. If electrodes are placed over the chest wall and the electrical impulse travels toward it, the recording of the electrode shows a positive or upward deflection (of course, the electrode has to be positive). When the impulse moves away from the electrode, it shows a negative deflection. Typically, the electrical activity during each contraction of the heart is recorded as a series of positive and negative waves, with each part of the wave representing im-pulse conduction along different parts of the heart.
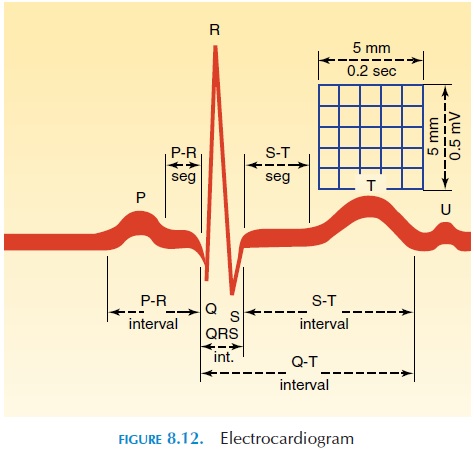
The first, small, positive upward deflection, the Pwave, represents the depolarization in the atria. Thecomplex of negative and large, positive deflection QRS complexrepresents the depolarization of theventricle. The repolarization activity of the atria oc- curs at the same time as the QRS complex. The last, positive upward deflection, theT wave, represents the repolarization of the ventricle. The distance be-tween the beginning of the P wave and the QRS com-plex—the PR interval—indicates the time taken for the impulse to travel from the atria to the ventricle.
Because the recording device, such as recording paper, moves at a standard speed, the distance be-tween the different waves indicates the duration taken for the impulse to travel from one region to an-other. Also, the height of the waves indicates the size of the muscle tissue (i.e., hypertrophied muscle tends to produce a larger wave). By placing the electrodes in different standard parts of the chest wall, arms, and legs, the electrical activity of the heart can be captured in various “viewpoints”—from the side, from above, or from below. Typically, electrodes are placed on the arms and legs and at six chest posi-tions. The instrument used to record the electrical changes is called an electrocardiograph.
A lot of information can be obtained by analyzing an ECG. Irregularities in heart rate, size of different chambers, location of pacemaker, presence and loca-tion of damaged or dead cardiac tissue, rate of con-duction, and conduction defects are just a few condi-tions that can be detected and diagnosed.
Related Topics