Chapter: Basic & Clinical Pharmacology : Antiseizure Drugs
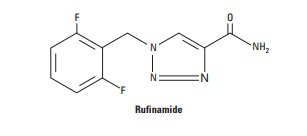
Rufinamide
RUFINAMIDE
Rufinamide
is a triazole derivative with little similarity to other antiseizure drugs.
Mechanism of Action
Rufinamide
is protective in the maximal electroshock and penty-lenetetrazol tests in rats
and mice. It decreases sustained high-frequency firing of neurons in vitro and
is thought to prolong the inactive state of the Na+ channel.
Significant interactions with GABA systems or metabotropic glutamate receptors
have not been seen.
Clinical Uses
Rufinamide
is approved in the USA for adjunctive treatment of sei-zures associated with
the Lennox-Gastaut syndrome in patients age 4 years and older. The drug is
effective against all seizure types in this syndrome and specifically against
tonic-atonic seizures. Recent data also suggest it may be effective against
partial seizures. Treatment in children is typically started at 10 mg/kg/d in
two equally divided doses and gradually increased to 45 mg/kg/d or 3200 mg/d,
which-ever is lower. Adults can begin with 400–800 mg/d in two equally divided
doses up to a maximum of 3200 mg/d as tolerated. The drug should be given with
food. The most common adverse events are somnolence, vomiting, pyrexia, and
diarrhea.
Pharmacokinetics
Rufinamide
is well absorbed, but plasma concentrations peak between 4 and 6 hours. The
half-life is 6–10 hours, and minimal plasma protein binding is observed.
Although cytochrome P450 enzymes are not involved, the drug is extensively
metabolized to inactive products. Most of the drug is excreted in the urine; an
acid metabolite accounts for about two thirds of the dose. In one study,
rufinamide did not appear to significantly affect the plasma concentrations of
other drugs used for the Lennox-Gastaut syn-drome such as topiramate,
lamotrigine, or valproic acid, but con-flicting data suggest more robust
interactions with other AEDs, including effects on rufinamide levels,
especially in children.
Related Topics