Chapter: Biotechnology Applying the Genetic Revolution: Genomics and Gene Expression
Radiation Hybrid and Cytogenetic Mapping
RADIATION
HYBRID AND CYTOGENETIC MAPPING
Library clones can sometimes
be unreliable because large cloned segments may actually consist of two
fragments of DNA, from different parts of the genome, inserted into the same
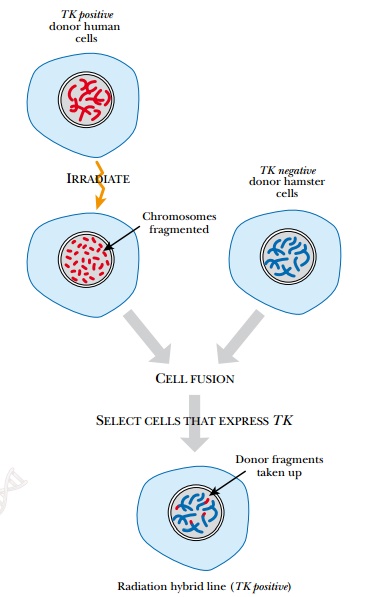
vector. Radiation hybrid mapping
overcomes these limitations by examining STSs or ESTs on original chromosomal
fragments (Fig. 8.7). To generate these, cultured human cells are treated with
x-rays or γ-rays to fragment the
chromosomes. The radiation dosage controls how often the chromosome breaks, and
thus the average length of the fragments. The human cells possess a marker
enzyme that allows them to grow on selective media. After irradiation, the
human cells are fused to cultured hamster cells using polyethylene glycol or
Sendai virus. The hamster cells do not have the selective marker. Consequently,
only those hamster cells that fuse with human cells survive. The fragments of
human chromosomes become part of the hamster nucleus, and the individual hybrid
cell lines can be examined by STS or EST mapping. Because the average fragment
length is known, these maps determine relative distance between two markers.

Cytogenetic mapping is
another physical technique that uses original chromosomes. When chromosomes are
placed on microscope slides and stained, they form banding patterns that are
visible under a light microscope. This cytogenetic
map shows where a gene or marker lies relative to the stained bands (Fig.
8.8). Cytogenetic maps are very low resolution compared with the other mapping
techniques, yet they are useful to compare gene locations on a large scale.

Related Topics