Chapter: Clinical Anesthesiology: Anesthetic Management: Anesthesia for Patients with Cardiovascular Disease
Mitral Regurgitation
MITRAL REGURGITATION
Preoperative Considerations
Mitral regurgitation can develop acutely
or insidiously as a result of a large number of disorders. Chronic mitral
regurgitation is usually the result of rheumatic fever (often with concomitant
mitral stenosis); con-genital or developmental abnormalities of the valve
apparatus; or dilatation, destruction, or calcification of the mitral annulus.
Acute mitral regurgitation is usually due to myocardial ischemia or infarction
(papillary muscle dysfunction or rupture of a chorda tendinea), infective
endocarditis, or chest trauma.
Pathophysiology
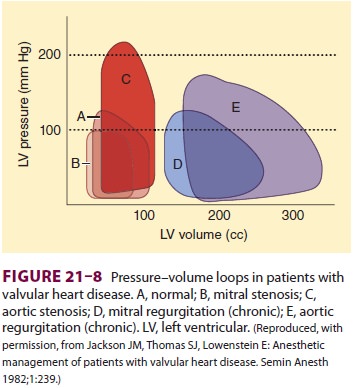
The principal derangement is a reduction
in forward stroke volume due to backward flow of blood into the left atrium
during systole. The left ventricle compen-sates by dilating and increasing
end-diastolic volume (Figure 21–8). Regurgitation thorugh the mitral valve
initially maintains a normal end systolic volume in spite of an increased end
diastolic volume. However, as the disease progresses the end systolic volume
increases. By increasing end-diastolic volume, the volume-overloaded left
ventricle can maintain a normal cardiac output despite blood being ejected
retrograde into the atrium. With time, patients with chronic mitral
regurgitation eventually develop eccentric left ventricular hypertrophy and
progres-sive impairment in contractility. In patients with severe mitral
regurgitation, the regurgitant volume may exceed the forward stroke volume. In
time, wall stress increases, resulting in an increased demand for myocardial
oxygen supply.
The regurgitant volume passing through
the mitral valve is dependent on the size of the mitral valve orifice (which
can vary with ventricular cav-ity size), the heart rate (systolic time), and
the left ventricular–left atrial pressure gradient during systole. The last
factor is af fected by the relative resistances of the two outflow paths from
the left ventricle, namely, SVR and left atrial compliance. Thus, a decrease in
SVR or an increase in mean left atrial pressure will reduce the regurgitant
volume. Atrial compliance also determines the predomi-nant clinical manifestations.
Patients with normal or reduced atrial compliance (acute mitral regurgita-tion)
have primarily pulmonary vascular congestion and edema. Patients with increased
atrial compli-ance (long-standing mitral regurgitation resulting in a large
dilated left atrium) primarily show signs of a reduced cardiac output. Most
patients are between the two extremes and exhibit symptoms of both pul-monary
congestion and low cardiac output. Patients with a regurgitant fraction of less
than 30% of the total stroke volume generally have mild symptoms. Regurgitant
fractions of 30% to 60% generally cause
moderate symptoms, whereas fractions
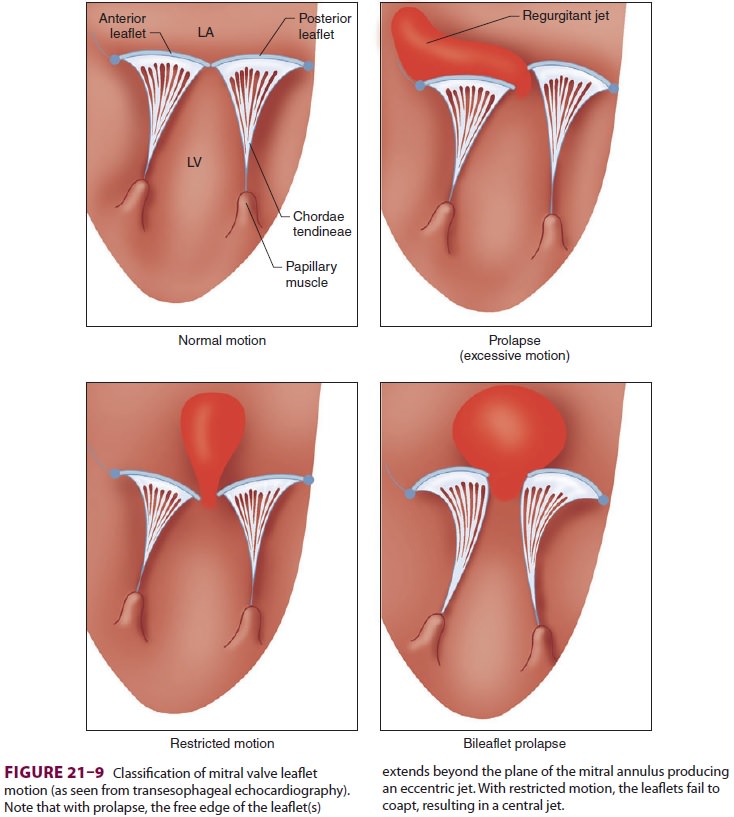
greater than 60% are associated with severe disease.Echocardiography,
particularly TEE, is useful in delineating the underlying pathophysiology of
mitral regurgitation and guiding treatment. Mitral valve leaflet motion is
often described as normal, prolaps-ing, or restrictive (Figure 21–9). Excessive motion
or prolapse is defined by systolic movement of a leaflet beyond the plane of
the mitral valve and into the left atrium.
Calculating Regurgitant Fraction
To calculate regurgitant fraction (RF),
forward stroke volume (SV) and the regurgitant stroke vol-ume (RSV) must be
measured. Although they can both be estimated by catheterization data, pulsed
Doppler echocardiography provides reasonably acute calculations. Stroke volume
is measured at the left ventricular outflow tract (LVOT) and at the mitral
valve (MV), where
Stroke volume = cross-sectional area (A) × (TVI) and cross-sectional area (A) can be approximated as,
A = 0.785 × (diameter)2
The time–velocity integral (TVI) is the
integral of the velocity versus the time signal obtained with pulsed Doppler.
The TVI reflects the distance the blood has traveled during a heart beat. By
knowing the area through which the blood travels and the distance traveled, it
is possible to estimate the stroke volume. This is the case because the area is
expressed in centimeters squared, and the distance is expressed in centimeters.
The product of these measures is cubic centimeters or milliliters—hence, the
stroke volume for each heartbeat.
Thus, the volume of blood that enters
through the mitral valve must be the same as that passing through the left
ventricular outflow track. Any dif-ference between the two represents the
amount of the volume that initially entered the left ventricle, but that did
not pass the LVOT. This is the volume that regurgitated into the left atrium.
RSVmitral regurgitation =(AMV ×VTIMV)− (ALVOT
× TVILVOT),
and
RF = RSV/SV
An RSV greater than 65 mL usually
correlates with severe mitral regurgitation.
Treatment
Afterload reduction is beneficial in
most patients and may even be lifesaving in patients with acute mitral
regurgitation. Reduction of SVR increases forward SV and decreases the
regurgitant volume. Surgical treatment is usually reserved for patients with
moderate to severe symptoms. Valvuloplasty or valve repair are performed
whenever possible to avoid the problems associated with valve replace-ment (eg,
thromboembolism, hemorrhage, and prosthetic failure). Catheter-mediated valve
repairs are continually being refined, potentially reduc-ing the need for
“open” surgery. Anesthesiologists skilled in advanced perioperative
echocardiogra-phy assist in correctly identifying the leaflet(s) to be repaired
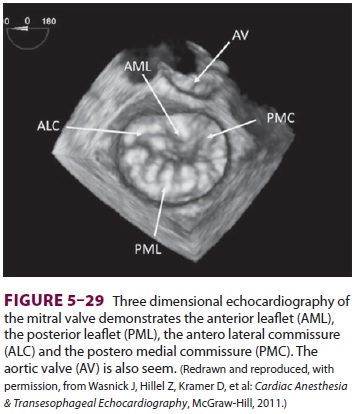
and determining the repair’s success. Three-dimensional echocardiography is
increas-ingly employed to assist in the assessment of the mitral valve (see
Figure 5-29).
Anesthetic Management
A. Objectives
Anesthetic management should be tailored
to the severity of mitral regurgitation as well asthe underlying left
ventricular function. Factors that exacerbate the regurgitation, such as slow
heart rates and acute increases in afterload, should be avoided. Bradycardia
can increase the regurgitant volume by increasing left ventricular
end-diastolic volume and acutely dilating the mitral annulus. The heart rate
should ideally be kept between 80 and 100 beats/min. Acute increases in left
ventricular afterload, such as with endotracheal intubation and surgical
stimula-tion under “light” anesthesia, should be treated rap-idly but without
excessive myocardial depression. Excessive volume expansion can also worsen the
regurgitation by dilating the left ventricle.
B. Monitoring
Monitors are based on the severity of
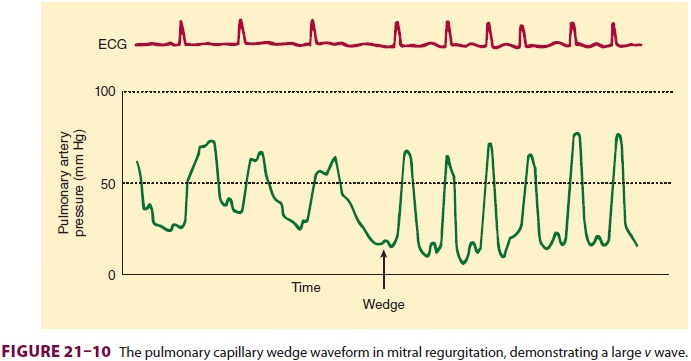
ventricular dysfunction, as well as the procedure. Mitral regur-gitation may be
recognized on the pulmonary artery wedge waveform as a large v wave and a rapid y descent (Figure 21–10). The height of the v wave is inversely related to atrial
and pulmonary vascular compliance, but is directly proportional to pulmo-nary
blood flow and the regurgitant volume; thus, the v wave may not be prominent in patients with chronic mitral
regurgitation, except during acute deterioration. Very large v waves are often apparent on the
pulmonary artery pressure waveform, even without wedging the catheter.
Color-flow Doppler TEE can be invaluable in quantitating the severity of
the regurgitation and guiding
therapeutic interven-tions in patients with severe mitral regurgitation. By
definition, blood flow reverses in the pulmonary veins during systole with
severe mitral regurgitation.
C. Choice of Agents
Patients with relatively well-preserved
ventricular function tend to do well with most anesthetic tech-niques. Spinal
and epidural anesthesia are well toler-ated, provided bradycardia is avoided.
Patients with moderate to severe ventricular impairment may be sensitive to
depression from high concentrations of volatile agents. An opioid-based
anesthetic may be more suitable for those patients—again, provided bradycardia
is avoided.
Related Topics