Chapter: Clinical Anesthesiology: Anesthetic Management: Anesthesia for Patients with Cardiovascular Disease
Ischemic Heart Disease
ISCHEMIC HEART DISEASE
Preoperative Considerations
Myocardial ischemia is characterized by
a meta-bolic oxygen demand that exceeds the oxygen sup-ply. Ischemia can
therefore result from a marked increase in myocardial metabolic demand, a
reduc-tion in myocardial oxygen delivery, or a combina-tion of both. Common
causes include coronary arterial vasospasm or thrombosis; severe hyperten-sion
or tachycardia (particularly in the presence of ventricular hypertrophy);
severe hypotension, hypoxemia, or anemia; and severe aortic stenosis or
regurgitation.
By far, the most common cause of
myocardial ischemia is atherosclerosis of the coronary arteries. CAD is responsible
for about 25% of all deaths in Western societies and is a major cause of
periop-erative morbidity and mortality. The overall inci-dence of CAD in
surgical patients is estimated to be between 5% and 10%. Major risk factors for
CAD include hyperlipidemia, hypertension, diabetes, cigarette smoking,
increasing age, male sex, and a positive family history. Other risk factors
includeobesity, a history of cerebrovascular or peripheral vascular disease,
menopause, use of high-estrogen oral contraceptives (in women who smoke), and a
sedentary lifestyle.
CAD may be clinically manifested by
symp-toms of myocardial necrosis (infarction), ischemia (usually angina),
arrhythmias (including sudden death), or ventricular dysfunction (congestive
heart failure). When symptoms of congestive heart failure predominate, the term
“ischemic cardiomyopathy” is often used.
Unstable Angina
Unstable angina is defined as (1) an
abrupt increase in severity, frequency (more than three episodes per day), or
duration of anginal attacks (crescendo angina): (2) angina at rest; or (3) new
onset of angina (within the past 2 months) with severe or frequent episodes
(more than three per day). Unstable angina may occur following MI or be
precipitated by non-cardiac medical conditions (including severe ane-mia,
fever, infections, thyrotoxicosis, hypoxemia, and emotional distress) in
previously stable patients.
Unstable angina, particularly when it is
asso-ciated with significant ST-segment changes at rest, usually reflects
severe underlying coronary disease and frequently precedes MI. Plaque
disruption with platelet aggregates or thrombi and vasospasm are fre-quent
pathological correlates. Critical stenosis in one or more major coronary
arteries is present in more than 80% of patients with these symptoms. Patients
with unstable angina require evaluation and treat-ment, which may include
admission to a coronary care unit and some form of coronary intervention.
Chronic Stable Angina
Anginal chest pains are most often
substernal, exer-tional, radiating to the neck or arm, and relieved by rest or
nitroglycerin. Variations are common, including epigastric, back, or neck pain,
or transient shortness of breath from ventricular dysfunction (anginal
equivalent). Nonexertional ischemia and silent (asymptomatic) ischemia are
recognized as fairly common occurrences. Patients with diabetes have an
increased incidence of silent ischemia.
Symptoms are generally absent until the
ath-erosclerotic lesions cause 50% to 75% occlusion of the coronary
circulation. When a stenotic segment reaches 70% occlusion, maximum
compensatory dilatation is usually present distally: blood flow is generally
adequate at rest, but becomes inadequate with increased metabolic demand. An
extensive col-lateral blood supply allows some patients to remain relatively
asymptomatic in spite of severe disease. Coronary vasospasm is also a cause of
transient transmural ischemia in some patients; 90% of vaso-spastic episodes
occur at preexisting stenotic lesions in epicardial vessels and are often
precipitated by a variety of factors, including emotional upset and
hyperventilation (Prinzmetal’s angina). Coronary spasm is most often observed
in patients who have angina with varying levels of activity or emotional stress
(variable-threshold); it is least common with classic exertional
(fixed-threshold) angina.
The overall prognosis of patients with
CAD is related to both the number and severity of coronary obstructions, as
well as to the extent of ventricular dysfunction.
Treatment of Ischemic Heart Disease
The general approach in treating
patients with isch-emic heart disease is five-fold:
·
Correction
of risk factors, with the hope of slowing disease progression.
·
Modification
of the patient’s lifestyle to reduce stress and improve exercise tolerance.
·
Correction
of complicating medical conditions that can exacerbate ischemia (ie,
hypertension, anemia, hypoxemia, hyperthyroidism, fever, infection, or adverse
drug effects).
·
Pharmacological
manipulation of the myocardial oxygen supply–demand relationship.
·
Correction
of coronary lesions by percutaneous coronary intervention (angioplasty [with or
without stenting] or atherectomy) or coronary artery bypass surgery.
The last three approaches are of direct
relevance to anesthesiologists. The same principles should be applied in the
care of these patients in both the oper-ating room and the intensive care unit.
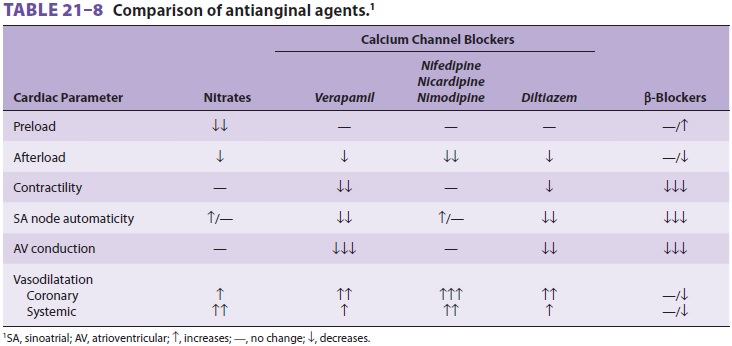
The most commonly used pharmacological
agents are nitrates, β-blockers, and calcium channel blockers.
These drugs also have potent circulatory effects, which are compared in Table 21–8.
Any of these agents can be used for mild angina. Calcium channel blockers are
the drugs of choice for patients with predominantly vasospastic angina. β-Blockers
improve the long-term outcome of patients with CAD. Nitrates are good agents
for both types of angina.
A. Nitrates
Nitrates relax all vascular smooth
muscle, but have a much greater effect on venous than on arterial vessels.
Decreasing venous and arteriolar tone and reducing the effective circulating
blood volume (cardiac pre-load) reduce wall tension afterload. These effects
tend to reduce myocardial oxygen demand. The prominent venodilatation makes
nitrates excellent agents when congestive heart failure is also present.
Perhaps equally important, nitrates
dilate the coronary arteries. Even minor degrees of dilatation at stenotic
sites may be sufficient to increase blood flow, because flow is directly
related to the fourth power of the radius. Nitrate-induced coronary vaso-dilatation
preferentially increases subendocardial blood flow in ischemic areas. This
favorable redistri-bution of coronary blood flow to ischemic areas maybe
dependent on the presence of collaterals in the coronary circulation.Nitrates
can be used for both the treatment of acute ischemia and prophylaxis against
frequent anginal episodes. Unlike β-blockers and calcium channel blockers,
nitrates do not have a negative inotropic effect—a desirable feature in the
presence of ventricular dysfunction. Intravenous nitroglyc-erin can also be
used for controlled hypotensive anesthesia.
B. Calcium Channel Blockers
The effects and uses of the most
commonly used calcium channel blockers are shown in Table 21–9. Calcium channel
blockers reduce myocardial oxy-gen demand by decreasing cardiac afterload and
augment oxygen supply by increasing blood flow (coronary vasodilatation).
Verapamil and diltiazem also reduce demand by slowing the heart rate.
Nifedipine’s potent eff ects on the
systemic blood pressure may precipitate hypotension, reflex tachycardia, or
both; its fast-onset preparations (eg, sublingual) have been associated with MI
in some patients. Its tendency to decrease afterload generally offsets any
negative inotropic effect. The slow-release form of nifedipine is associated
with much less reflex tachycardia and is more suitable than other agents for
patients with ventricular
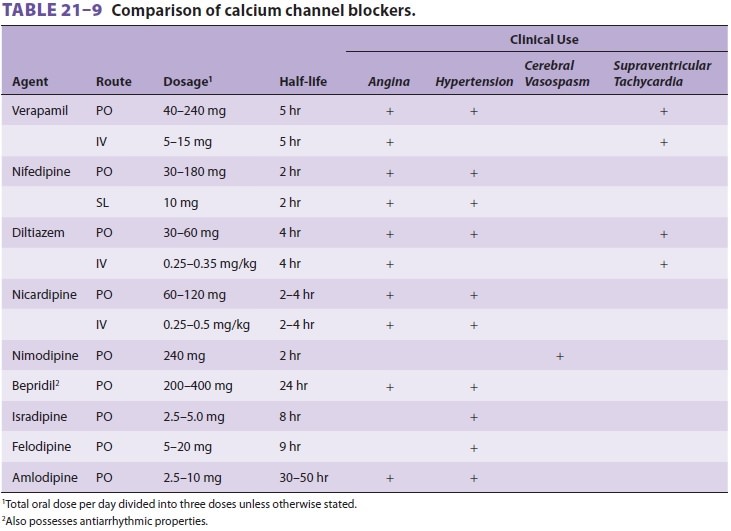
dysfunction. In contrast, verapamil and
diltiazem have greater effects on cardiac contractility and atrioventricular
(AV) conduction and therefore should be used cautiously, if at all, in patients
with ventricular dysfunction, conduction abnormalities, or bradyarrhythmias.
Diltiazem seems to be better tolerated than verapamil in patients with impaired
ventricular function. Nicardipine, nimodipine, and clevidipine generally have
the same effects as nife-dipine; nimodipine is primarily used in preventing
cerebral vasospasm following subarachnoid hemor-rhage, whereas nicardipine is
used as an intravenous arterial vasodilator. Clevidipine is an
ultrashort–act-ing arterial vasodilator.
Calcium channel blockers can have
significant interactions with anesthetic agents. All calcium channel blockers
potentiate both depolarizing and nondepolarizing neuromuscular blocking agents
and the circulatory effects of volatile agents. Both verapamil and diltiazem
can potentiate depression of cardiac contractility and conduction in the AV
node by volatile anesthetics. Nifedipine and simi-lar agents can potentiate systemic
vasodilatation by volatile and intravenous agents.
C. β-Adrenergic Blocking Agents
Th ese drugs decrease myocardial oxygen
demand by reducing heart rate and contractility, and, in some cases, afterload
(via their antihypertensive effect). Optimal blockade results in a resting
heart rate between 50 and 60 beats/min and prevents appreciable increases with
exercise (<20 beats/min increase during exercise).
Available agents differ in receptor selectivity, intrinsic sympathomimetic
(partial agonist) activity, and membrane-stabiliz-ing properties ( Table 21–10).
Membrane stabili-zation, often described as a quinidine-like effect, results in
antiarrhythmic activity. Agents with
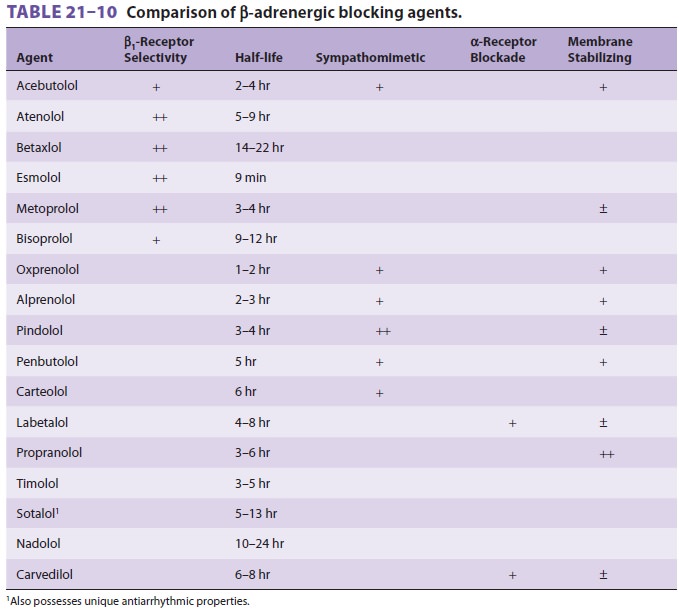
intrinsic sympathomimetic properties are
better tol-erated by patients with mild to moderate ventricu-lar dysfunction.
Certain β-blockers (carvedilol and extended-duration
metoprolol) improve survival in patients with chronic heart failure. This has
not been shown to be a drug class effect. Blockade of β2-adrenergic receptors also can mask hypoglyce-mic
symptoms in patients with diabetes, delay meta-bolic recovery from
hypoglycemia, and impair the handling of large potassium loads. Cardioselective
(β1-receptor-specific)
agents, although generally better tolerated than nonselective agents in
patients with reactive airways, must still be used cautiously in such patients.
The selectivity of cardioselective agents tends to be dose dependent. Patients
on long-standing β-blocker therapy should have these agents continued
perioperatively. Acute β-blocker withdrawal in the perioperative
period places patients at a markedly increased risk of cardiac mor-bidity and
mortality.
Documentation of avoidance of β-blocker
withdrawal is a frequent tool by which “quality” of anesthesia services can be
assessed by regulatory agencies.
D. Other Agents
ACE inhibitors prolong survival in
patients with congestive heart failure or left ventricular dysfunc-tion.
Chronic aspirin therapy reduces coronary events in patients with CAD and
prevents coronary
and ischemic cerebral events in at-risk
patients. Antiarrhythmic therapy in patients with complex ventricular ectopy
who have significant CAD and left ventricular dysfunction should be guided by
an electrophysiological study. Patients with inducible sustained ventricular
tachycardia (VT) or ventricu-lar fibrillation are candidates for an automatic
inter-nal cardioverter-defibrillator (ICD). Treatment of ventricular ectopy
(with the exception of sustained VT) in patients with good ventricular function
does not improve survival and may increase mortality. In contrast, ICDs have
been shown to improve survival in patients with advanced cardiomyopathy
(ejection fraction <30%), even in the absence of demonstrable
arrhythmias.
E. Combination Therapy
Moderate to severe angina frequently
requires com-bination therapy with two or all three classes of agents. Patients
with ventricular dysfunction may not tolerate the combined negative inotropic
effect of a β-blocker and a calcium channel blocker together; an
ACE inhibitor is better tolerated and seems to improve survival. Similarly, the
additive effect of a β-blocker and a calcium channel blocker
on the AV node may precipitate heart block in susceptible patients.
Related Topics