Chapter: Biochemical Pharmacology : G protein-coupled receptors
False transmitters
'False
transmitters'
Another drug closely similar
to DOPA but used for differ-ent applications is α-methyl-DOPA (Figure 10.19a). This molecule
acts in the peripheral autonomous system but also enters the brain, by the same
route as DOPA. It is converted by DOPA decarboxylase to the `false transmitter'α-methyl-dopamine. Like dopamine or norepinephrine, α-methyl-dopamine is accumulated inside the transmitter vesicles,
and released in response to action potentials. While it has no strong effect on
postsynaptic α1-receptors,
it does ac-tivate α2-receptors. It will therefore inhibit the further re-lease of transmitter
without stimulating the postsynaptic neuron. The effect of methyl-DOPA is
augmented by the fact that it is fairly resistant to monoamine oxidase. Its
mode of action resembles that of clonidine (which accom-plishes the same in a
less roundabout manner).
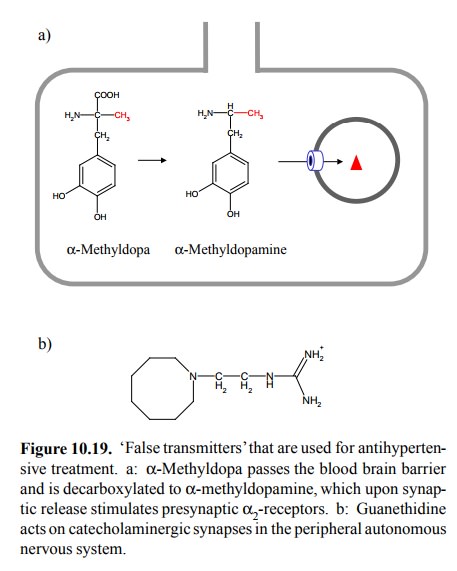
Another
example of a `false transmitter' is the drug guanethidine (Figure 10.19b). This
drug is quite different in structure from methyl-DOPA. It is therefore not
piggy-backed across the blood brain barrier by the aromatic amino acid
transporter (nor by any other specific transporter), and since it is polar it
does not cross by simple diffusion either. However, it may enter
post-ganglionic sympathetic neurons in the periphery by way or reuptake
transporters. Accord-ingly, guanethidine can lower blood pressure by a purely
peripheral mechanism.
Related Topics