Chapter: Biochemical Pharmacology : G protein-coupled receptors
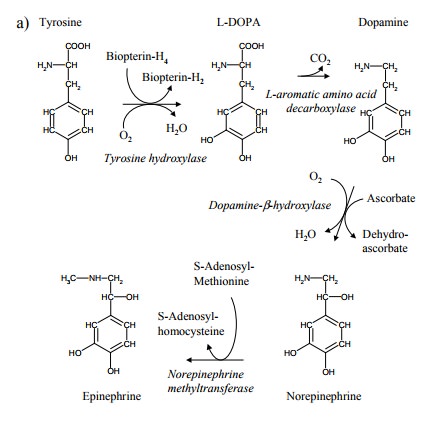
Biosynthesis and degradation of catecholamines
Biosynthesis and degradation
of catecholamines
The catecholamines -
dopamine, norepinephrine, and epinephrine are successively derived from
tyrosine. Syn-thesis occurs in the nerve terminals and in the adrenal gland.
Tyrosine hydroxylase catalyzes the first step (Figure 10.2a) and is the major
site of regulation (inhibition by dopamine and noradrenaline, activation by
cAMP). This step gives rise to 3,4-dihydroxyphenylalanine (L-DOPA), which in
turn is a substrate for L-aromatic acid decarboxylase. Decarboxylation yields
the first mediator, dopamine. The sub-strate specificity of the decarboxylase
is rather low, and it thus will also accept 5-hydroxytryptophan (the precursor
of serotonin, Figure 10.2a) and a variety of synthetic analogs, as we shall see
later. Further hydroxylation of dopamine leads to norepinephrine, and
methylation to epinephrine.
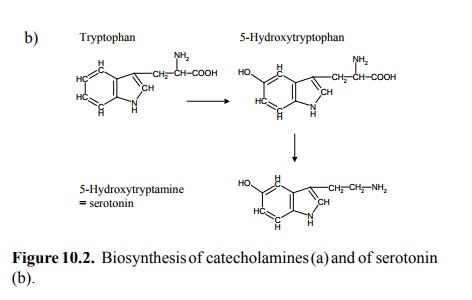
The biosynthesis of serotonin
is similar to that of dopamine and also involves enzymatic hydroxylation and
subsequent decarboxylation (Figure 10.2b).
Catecholamines and serotonin
also share the major mode of degradation. Both are substrates for monoamine
oxidase (MAO), although one subtype of this enzyme (MAO A) has a preference for
catecholamines, whereas the other (MAO
B) acts more efficiently on serotonin. MAO is found both within the
synaptic terminals, where it degrades any excess transmitter, and in the liver,
where it scavenges circulating catecholamines.
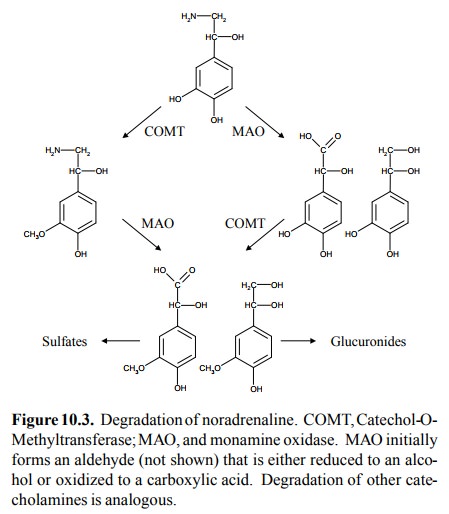
A second enzyme,
catechol-O-methyltransferase (COMT), inactivates catecholamines by attaching a
methyl group to the 3-OH group of the catechol moiety (Figure 10.3). This is
illustrated here only for norepinephrine but hap-pens in just the same way for
the other catecholamines as well. Since both MAO and COMT have fairly loose
sub-strate specificities, they can operate on each other's prod-ucts and hence
in either sequence. Before elimination, the MAO/COMT-generated products may or
may not be fur-ther conjugated by sulfation or glucuronidation, by the same
enzymes that also conjugate drug molecules.
Related Topics