Chapter: Basic & Clinical Pharmacology : Antihypertensive Agents
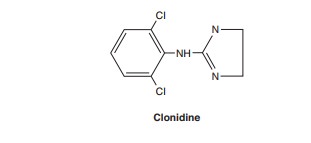
Clonidine
CLONIDINE
Blood pressure lowering by clonidine results from reduction of cardiac output due to decreased heart rate and relaxation of capaci-tance vessels, as well as a reduction in peripheral vascular resistance. Reduction in arterial blood pressure by clonidine is accompa-nied by decreased renal vascular resistance and maintenance of renal blood flow. As with methyldopa, clonidine reduces blood pressure in the supine position and only rarely causes postural hypotension. Pressor effects of clonidine are not observed after ingestion of therapeutic doses of clonidine, but severe hyperten-sion can complicate a massive overdose.
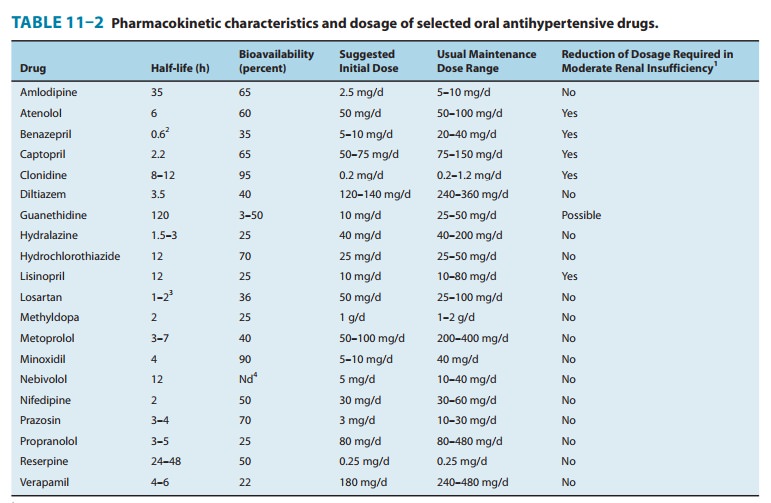
Pharmacokinetics & Dosage
Typical
pharmacokinetic characteristics are listed in Table 11–2. Clonidine is
lipid-soluble and rapidly enters the brain from thecirculation. Because of its
relatively short half-life and the fact that its antihypertensive effect is
directly related to blood concentration, oral clonidine must be given twice a
day (or as a patch, below) to maintain smooth blood pressure control. However,
as is not the case with methyldopa, the dose-response curve of clonidine is
such that increasing doses are more effective (but also more toxic).
A
transdermal preparation of clonidine that reduces blood pressure for 7 days
after a single application is also available. This preparation appears to
produce less sedation than clonidine tablets but is often associated with local
skin reactions.
Toxicity
Dry
mouth and sedation are common. Both effects are centrally mediated and
dose-dependent and coincide temporally with the drug’s antihypertensive effect.
Clonidine
should not be given to patients who are at risk for mental depression and
should be withdrawn if depression occurs during therapy. Concomitant treatment
with tricyclic antidepres-sants may block the antihypertensive effect of
clonidine. The interaction is believed to be due to α-adrenoceptor–blocking actions of the
tricyclics.
Withdrawal
of clonidine after protracted use, particularly with high dosages (more than 1
mg/d), can result in life-threatening hypertensive crisis mediated by increased
sympathetic nervous activity. Patients exhibit nervousness, tachycardia,
headache, and sweating after omitting one or two doses of the drug. Because of
the risk of severe hypertensive crisis when clonidine is suddenly withdrawn,
all patients who take clonidine should be warned of the possibility. If the
drug must be stopped, it should be done gradually while other antihypertensive
agents are being substi-tuted. Treatment of the hypertensive crisis consists of
reinstitution of clonidine therapy or administration of α- and β-adrenoceptor–
blocking agents.
Related Topics