Chapter: Basic & Clinical Pharmacology : Antihypertensive Agents
Angiotensin-Converting Enzyme (ACE) Inhibitors
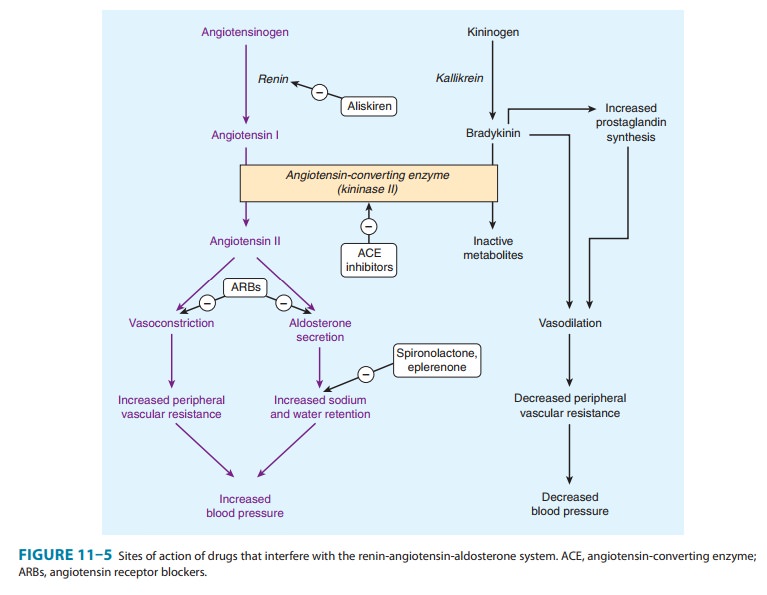
ANGIOTENSIN-CONVERTING ENZYME
(ACE) INHIBITORS
Captopril and other drugs in this class inhibit the
convertingenzyme peptidyl dipeptidase that hydrolyzes angiotensin I to
angiotensin II and (under the name plasma kininase) inactivatesbradykinin, a
potent vasodilator, which works at least in part by stimulating release of
nitric oxide and prostacyclin. The hypoten-sive activity of captopril results
both from an inhibitory action on the renin-angiotensin system and a
stimulating action on the kallikrein-kinin system (Figure 11–5). The latter
mechanism has been demonstrated by showing that a bradykinin receptor
antag-onist, icatibant , blunts the
blood pressure-lowering effect of captopril.
Enalapril is an oral prodrug that is converted by
hydrolysis toa converting enzyme inhibitor, enalaprilat, with effects similar
to those of captopril. Enalaprilat itself is available only for intrave-nous
use, primarily for hypertensive emergencies. Lisinopril is a lysine derivative
of enalaprilat. Benazepril, fosinopril,
moexipril,perindopril, quinapril, ramipril, and trandolapril are otherlong-acting members of the class. All are
prodrugs, like enalapril, and are converted to the active agents by hydrolysis,
primarily in the liver.
Angiotensin
II inhibitors lower blood pressure principally by decreasing peripheral vascular
resistance. Cardiac output and heart rate are not significantly changed. Unlike
direct vasodilators, these agents do not result in reflex sympathetic
activation and can be used safely in persons with ischemic heart disease. The
absence of reflex tachycardia may be due to downward resetting of the
baroreceptors or to enhanced parasympathetic activity.
Although
converting enzyme inhibitors are most effective in conditions associated with
high plasma renin activity, there is no good correlation among subjects between
plasma renin activity and antihypertensive response. Accordingly, renin
profiling is unnecessary.
ACE
inhibitors have a particularly useful role in treating patients with chronic
kidney disease because they diminish pro-teinuria and stabilize renal function
(even in the absence of lower-ing of blood pressure). This effect is
particularly valuable in diabetes, and these drugs are now recommended in
diabetes even in the absence of hypertension. These benefits probably result
from improved intrarenal hemodynamics, with decreased glom-erular efferent
arteriolar resistance and a resulting reduction of intraglomerular capillary
pressure. ACE inhibitors have also proved to be extremely useful in the
treatment of heart failure, and after myocardial infarction, and there is
recent evidence that ACE inhibitors reduce the incidence of diabetes in
patients with high cardiovascular risk .
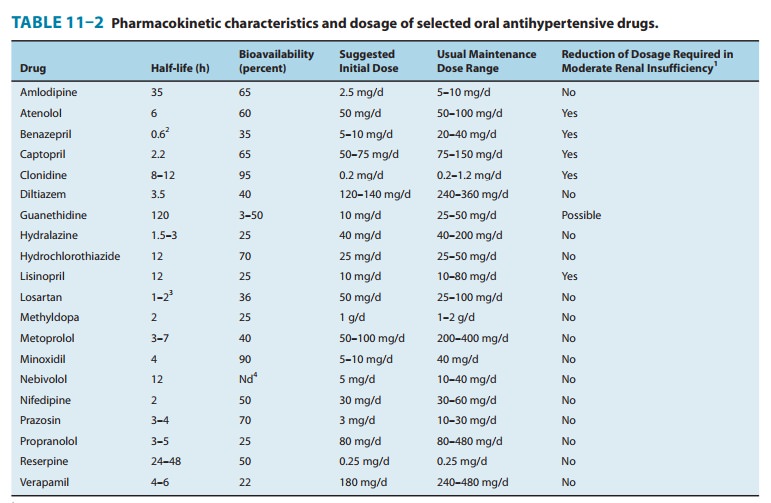
Pharmacokinetics & Dosage
Captopril’s
pharmacokinetic parameters and dosing recommenda-tions are set forth in Table
11–2. Peak concentrations of enalapri-lat, the active metabolite of enalapril,
occur 3–4 hours after dosing with enalapril. The half-life of enalaprilat is
about 11 hours. Typical doses of enalapril are 10–20 mg once or twice daily.
Lisinopril has a half-life of 12 hours. Doses of 10–80 mg once daily are
effective in most patients. All of the ACE inhibitors except fosinopril and
moexipril are eliminated primarily by the kidneys; doses of these drugs should
be reduced in patients with renal insufficiency.
Toxicity
Severe
hypotension can occur after initial doses of any ACE inhibitor in patients who
are hypovolemic as a result of diuretics, salt restriction, or gastrointestinal
fluid loss. Other adverse effects common to all ACE inhibitors include acute
renal failure (particularly in patients with bilateral renal artery stenosis or
stenosis of the renal artery of a solitary kidney), hyperkalemia, dry cough
sometimes accompanied by wheezing, and angio-edema. Hyperkalemia is more likely
to occur in patients with renal insufficiency or diabetes. Bradykinin and
substance P seem to be responsible for the cough and angioedema seen with ACE
inhibition.
ACE
inhibitors are contraindicated during the second and third trimesters of
pregnancy because of the risk of fetal hypoten-sion, anuria, and renal failure,
sometimes associated with fetal malformations or death. Recent evidence also
implicates first-trimester exposure to ACE inhibitors in increased teratogenic
risk. Captopril, particularly when given in high doses to patients with renal
insufficiency, may cause neutropenia or proteinuria. Minor toxic effects seen
more typically include altered sense of taste, allergic skin rashes, and drug
fever, which may occur in up to 10% of patients.Important drug interactions
include those with potassium supplements or potassium-sparing diuretics, which
can result in hyperkalemia. Nonsteroidal anti-inflammatory drugs may impair the
hypotensive effects of ACE inhibitors by blocking bradykinin-mediated
vasodilation, which is at least in part, prostaglandin mediated.
Related Topics